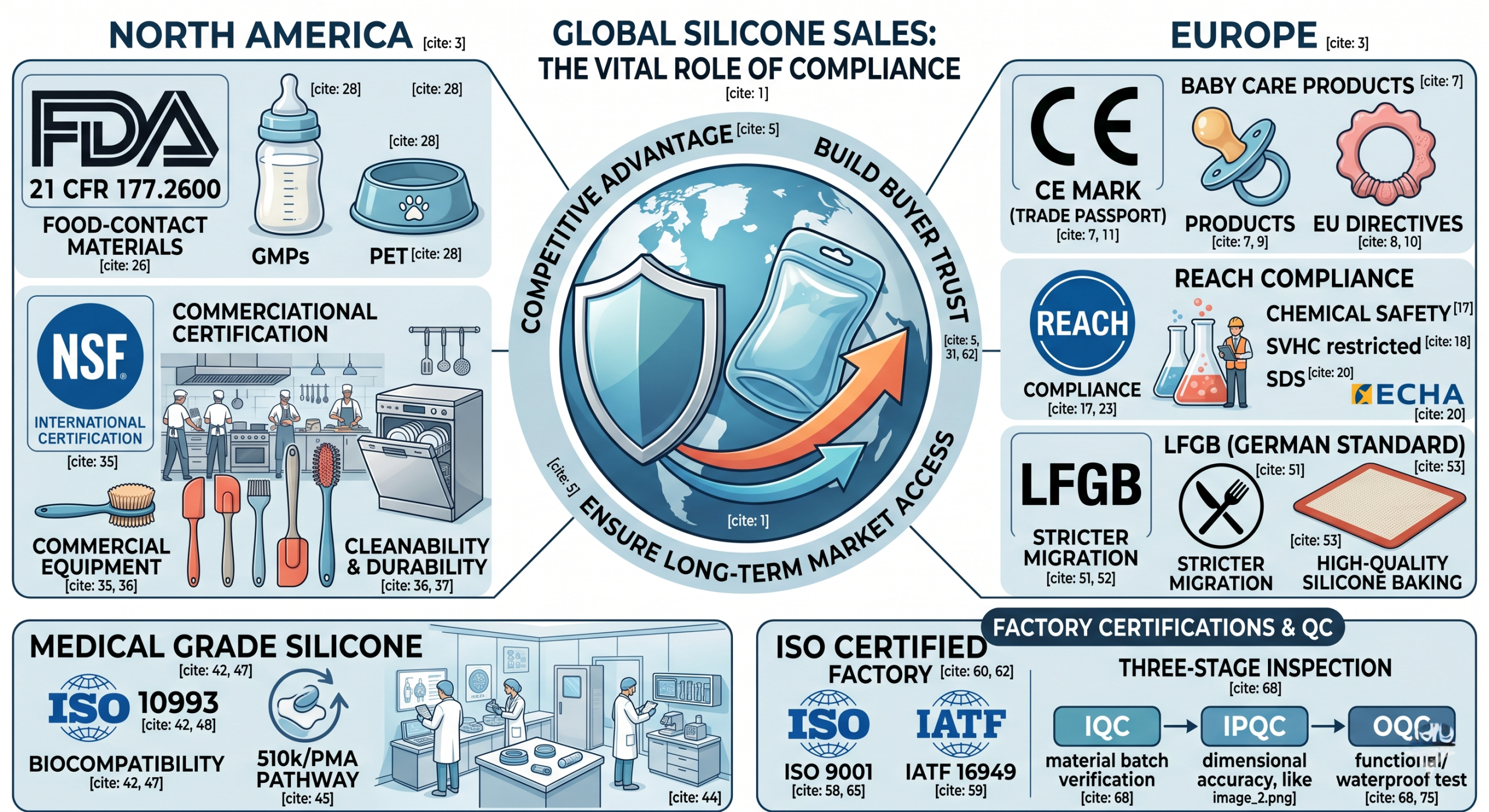
The risks of non-compliant Silicone Products
Selling in international markets without proper documentation can lead to shipment seizures, heavy fines, and permanent brand damage. For B2B buyers, ensuring that Silicone Products meet regional safety laws is the first step in risk mitigation.
Understanding regional regulatory variations
While North America relies heavily on FDA and NSF standards, Europe operates under the CE and REACH frameworks. Navigating these requires a deep understanding of how material science intersects with local consumer protection laws.
Key Takeaway: Compliance is not just a legal hurdle but a competitive advantage that builds buyer trust and ensures long-term market access for your Silicone Products.

How does the CE mark affect European market entry?
Mandatory safety standards for baby care
For items like pacifiers or teething toys, the CE mark (Conformité Européenne) is a non-negotiable gateway. It signals that the product aligns with EU health, safety, and environmental protection legislation. You can learn more about specialized mother and baby care solutions that prioritize these standards.
Steps to verify CE authenticity
Verification involves identifying relevant directives, such as the General Product Safety Directive, and compiling technical documentation that includes risk assessments and laboratory test results.
Key Takeaway: The CE mark serves as a “trade passport” for the European Economic Area; without it, consumer-facing silicone goods cannot be legally sold.
Table 1: CE Marking Core Components
| Component | Description | Relevance |
|---|---|---|
| Directive Identification | Selecting the correct EU law (e.g., Toy Safety) | High |
| Technical File | Documentation of design and testing | Mandatory |
| Declaration of Conformity | Manufacturer’s signed statement of compliance | Mandatory |
What does REACH compliance mean for material safety?
Managing chemical substances in production
REACH (Registration, Evaluation, Authorisation, and Restriction of Chemicals) focuses on the chemical building blocks of Silicone Products. It ensures that no “Substances of Very High Concern” (SVHC) are present in concentrations that could harm human health or the environment.
Why transparency in the supply chain matters
Achieving REACH compliance requires a meticulous audit of the raw material supply chain. Manufacturers must provide Safety Data Sheets (SDS) to prove that every catalyst and pigment used in the molding process is registered with the European Chemicals Agency (ECHA).
Key Takeaway: REACH is about chemical transparency; it ensures that every ingredient in your silicone formulation is accounted for and safe for the end-user.

Table 2: REACH vs. CE Comparison
| Feature | REACH | CE Marking |
|---|---|---|
| Focus | Chemical substance safety | Overall product safety/function |
| Geography | European Union | European Economic Area |
| Primary Requirement | Substance registration (ECHA) | Technical documentation & marking |
What are the core FDA requirements for food-grade silicone?
Testing protocols for food-contact items
In the United States, the FDA (Food and Drug Administration) regulates materials that come into contact with food under 21 CFR 177.2600. This standard ensures that Silicone Products do not leach harmful substances into food or beverages. This is particularly critical for items like pet products, where durability and safety are paramount.
Maintaining compliance for Silicone Products
FDA compliance isn’t a one-time event; it requires ongoing adherence to Good Manufacturing Practices (GMP). This includes using only FDA-approved raw materials and maintaining clean production environments to prevent cross-contamination.
Key Takeaway: FDA approval is the “gold standard” for the North American food and beverage industry, serving as a critical trust indicator for both retailers and consumers.
Table 3: FDA 21 CFR 177.2600 Testing Criteria
| Test Type | Objective | Limit |
|---|---|---|
| Distilled Water Extractives | Measuring migration in aqueous food | < 20 mg/sq. inch |
| n-Hexane Extractives | Measuring migration in fatty food | < 175 mg/sq. inch |
Why is NSF certification essential for kitchen supplies?
Material safety in commercial kitchens
While FDA sets the baseline, NSF International (National Sanitation Foundation) provides specialized certification for commercial kitchen equipment. NSF/ANSI standards evaluate the cleanability, corrosion resistance, and structural integrity of Silicone Products used in professional food service.
Performance testing for durability
NSF testing goes beyond chemistry; it includes “use and abuse” testing to ensure the silicone doesn’t crack or harbor bacteria over thousands of cycles in a commercial dishwasher.
Key Takeaway: NSF certification is often a prerequisite for high-volume commercial contracts in North America, as it guarantees both safety and industrial-grade performance.

Table 4: NSF Certification Levels
| Standard | Application | Focus |
|---|---|---|
| NSF/ANSI 51 | Food Equipment Materials | Toxicology and cleanability |
| NSF/ANSI 61 | Drinking Water Components | Leachate levels in water systems |
Does medical-grade silicone require special certifications?
Biocompatibility and ISO 10993
Medical-grade silicone must pass rigorous biocompatibility testing, primarily the ISO 10993 series. These tests ensure the material does not cause toxic, inflammatory, or immune responses when in contact with human tissue. Expert medical-grade silicone parts manufacturers typically operate in Class 100,000 cleanrooms to maintain these standards.
Regulatory path for silicone implants
For Class II and III medical devices, the FDA requires a Pre-Market Notification (510k) or Approval (PMA). This involves extensive clinical data and a “Master File” (MAF) submitted by the silicone raw material supplier.
Key Takeaway: Medical certification is the most stringent tier of compliance, requiring specialized manufacturing environments and exhaustive biological validation.
Table 5: ISO 10993 Testing Tiers
| Tier | Test Type | Duration |
|---|---|---|
| Surface Contact | Cytotoxicity, Sensitization | Short-term |
| Implantable | Systemic Toxicity, Genotoxicity | Long-term |
How do LFGB standards differ from FDA requirements?
The “Knife and Fork” symbol explained
The LFGB (Lebensmittel-, Bedarfsgegenstände- und Futtermittelgesetzbuch) is the German standard for food-contact materials. Recognized by the “Knife and Fork” symbol, it is often considered stricter than FDA standards, particularly regarding sensory testing (smell and taste).
Why LFGB is preferred for premium European goods
Many high-end European retailers require LFGB certification over FDA because it involves more comprehensive migration testing. Using platinum-cured Silicone Products is often necessary to pass the stringent LFGB requirements without the risk of “blooming” or odor.
Key Takeaway: If you are targeting the premium German or French markets, LFGB certification is a superior marketing tool and safety guarantee compared to basic FDA compliance.

Table 6: FDA vs. LFGB Migration Limits
| Parameter | FDA Requirement | LFGB Requirement |
|---|---|---|
| Overall Migration | Standardized | Higher sensitivity |
| Volatile Organic Matter | Not strictly limited | < 0.5% |
Which ISO certifications should your manufacturer hold?
ISO 9001 vs. IATF 16949
A manufacturer’s foundation rests on its Quality Management System (QMS). ISO 9001 is the global standard for general quality, while IATF 16949 is required for automotive-grade components. Partnering with an ISO-certified factory ensures that every batch of Silicone Products is produced under controlled, repeatable processes.
Building trust with international buyers
ISO certifications prove that a factory has a structured approach to continuous improvement, document control, and employee training, which significantly reduces the risk of batch-to-batch inconsistency.
Key Takeaway: ISO certification is an indicator of operational maturity; it assures buyers that the factory has the systems in place to handle complex global compliance requirements.
Table 7: Essential Factory Certifications
| Certification | Scope | Buyer Benefit |
|---|---|---|
| ISO 9001 | General Quality Management | Consistent product quality |
| ISO 13485 | Medical Device Quality | Regulatory alignment for healthcare |
| ISO 14001 | Environmental Management | Sustainability compliance |
How is quality controlled during mass production?
IQC to OQC: A three-stage quality system
Compliance is maintained through rigorous inspection. Incoming Quality Control (IQC) verifies raw material certifications. In-Process Quality Control (IPQC) monitors molding parameters, and Outgoing Quality Control (OQC) performs final functional tests before shipment.
Reducing risk in global logistics
For Silicone Products destined for North America or Europe, packaging must also be compliant. This includes moisture-proof sealing and clear labeling that matches the certification documents provided to customs officials.
Key Takeaway: Real-world compliance happens on the factory floor; a robust three-stage inspection process is the only way to ensure that theoretical standards are met in every physical part.
Table 8: Production Quality Metrics
| Stage | Key Inspection Task | Tools Used |
|---|---|---|
| IQC | Material Batch Verification | Spectrometer / Hardness Tester |
| IPQC | Dimensional Accuracy | CCD Visual Inspection / Calipers |
| OQC | Functional/Waterproof Testing | Pressure Chambers / Pull Testers |
Frequently Asked Questions
Can I sell silicone products in the US with only CE certification?
No. While CE is highly respected, the US market specifically requires FDA compliance for food-contact items and potentially UL or FCC certifications for electronic silicone components.
What’s the best way to prove LFGB compliance to a German buyer?
Provide a formal Declaration of Compliance (DoC) and a test report from an accredited third-party laboratory (like SGS or TÜV) that specifically mentions LFGB 81.64 or 81.30.
Can I use the same silicone grade for both FDA and LFGB markets?
It is possible if you use a high-purity, platinum-cured silicone that meets the more stringent LFGB migration limits, as it will naturally exceed the basic FDA requirements.
What’s the best certification for silicone used in wearable tech?
For wearables, look for ISO 10993-5 (Cytotoxicity) and ISO 10993-10 (Skin Sensitization) to ensure the material is safe for prolonged skin contact.
Can I rely on my manufacturer’s material certifications alone?
While essential, it is best practice to perform periodic “spot checks” via third-party labs to ensure the finished product—not just the raw material—remains compliant after the heat and pressure of the molding process.