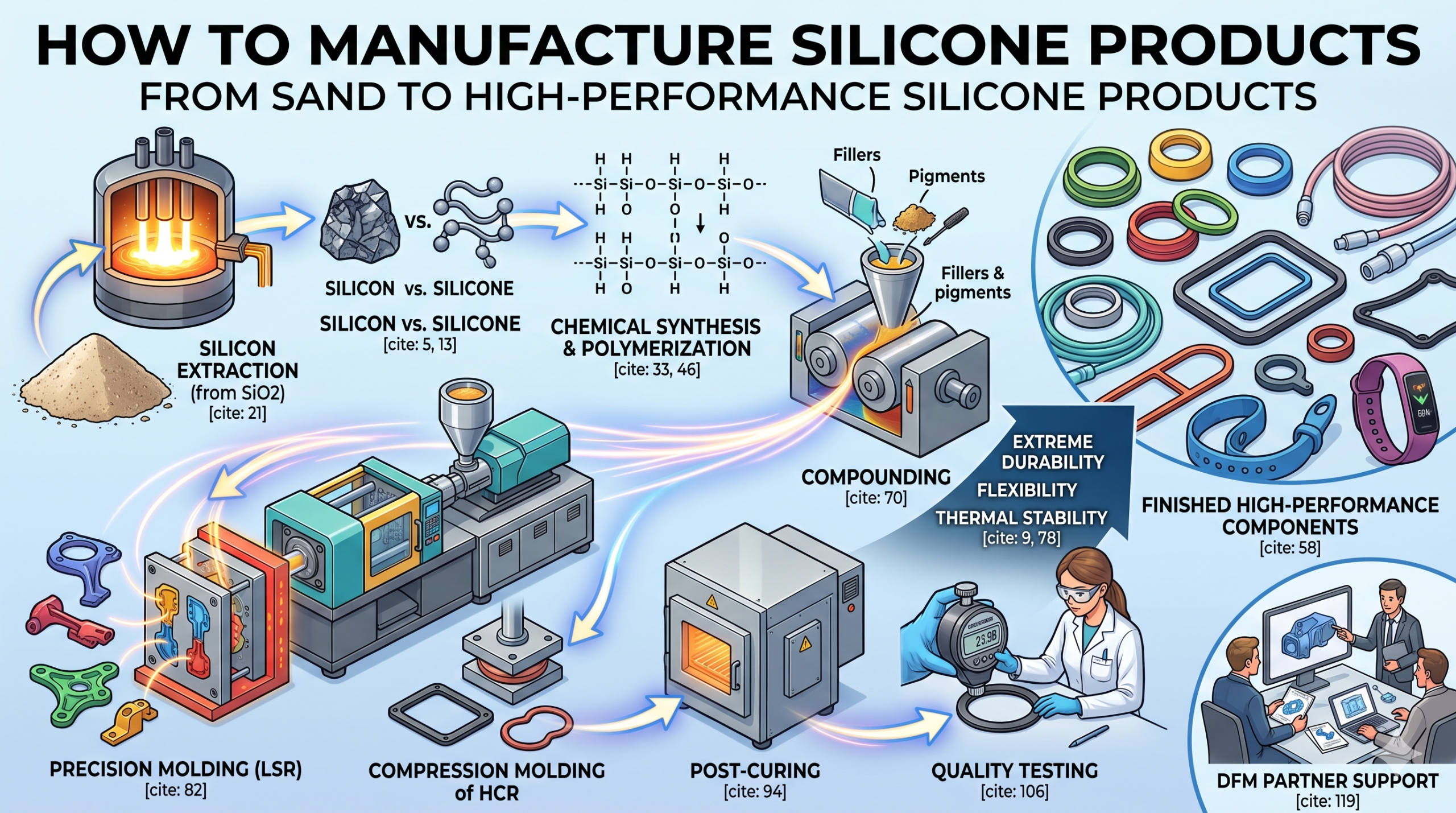
Silicone manufacturing transforms common sand into high-performance polymers through a sophisticated journey of thermal reduction, chemical synthesis, and precision molding. You might struggle with choosing the right materials for extreme environments where traditional plastics melt or crack under pressure. This inconsistency in material choice often leads to costly project delays and product failures in the field. Understanding the fundamental steps of how to manufacture silicone products provides the clarity needed to source durable, high-quality components for any application.
What exactly is silicone compared to silicon?

Silicone is a synthetic polymer made of silicon, oxygen, carbon, and hydrogen, whereas silicon is a natural chemical element found abundantly in the Earth’s crust. When you investigate how to manufacture silicone products, it is vital to distinguish between the raw element and the finished elastomer. Silicon serves as the inorganic foundation, while the addition of organic groups creates the flexible, heat-resistant material we use today.
The chemical foundations of polymers
The backbone of this material consists of alternating silicon and oxygen atoms, known as a siloxane link. This unique structure provides incredible thermal stability and flexibility across a wide range of temperatures.
Think about it:
- The Si-O bond is significantly stronger than the C-C bond found in organic rubbers.
- This strength prevents the material from degrading under intense UV light or ozone exposure.
- Organic side groups like methyl or phenyl are attached to the silicon atoms to modify properties.
Silicon vs. Silicone properties
Silicon is a hard, brittle crystalline solid used primarily in the semiconductor industry for electronic chips. In contrast, silicone is a versatile elastomer available in liquid, gel, or solid forms for industrial use.
Here is the deal:
- Silicon is a natural element (Si).
- Silicone is a man-made polymer (Polysiloxane).
- Silicon conducts electricity, while silicone typically acts as an excellent insulator.
Key Takeaway: Understanding the chemical difference ensures you select the correct material grade for high-performance industrial applications requiring flexibility and insulation.
| Attribute | Silicon (Element) | Silicone (Polymer) |
|---|---|---|
| Origin | Natural (from silica) | Synthetic (man-made) |
| State | Hard, brittle solid | Flexible elastomer/gel |
| Application | Semiconductors/Electronics | Medical/Automotive/Consumer |
This structural distinction defines why silicone is the preferred choice for seals and medical tubing over raw minerals.
How do we extract silicon from common silica?

Silicon extraction begins by heating high-purity quartz sand with carbon sources in an electric arc furnace at temperatures reaching 1,800 degrees Celsius. This high-energy phase is the first physical step in learning how to manufacture silicone products for global supply chains. The intense heat triggers a reduction reaction, stripping oxygen away from the silica to leave behind metallurgical-grade silicon.
Refining quartz sand at heat
The process requires massive amounts of electricity to maintain the extreme temperatures needed for the oxygen-carbon exchange. Carbon materials like coal or wood chips act as reducing agents during this violent chemical transformation.
But wait, there’s more:
- The oxygen in the quartz (SiO2) bonds with the carbon to form carbon monoxide gas.
- Molten silicon settles at the bottom of the furnace and is tapped off into molds.
- The resulting silicon is typically 98% to 99% pure before further chemical processing.
Key Takeaway: High-purity extraction is the essential baseline that determines the eventual clarity and strength of your finished silicone components.
| Process Step | Temperature | Primary Output |
|---|---|---|
| Furnace Input | Ambient | Quartz sand + Carbon |
| Reduction | 1,800°C | Molten Silicon |
| Cooling | Controlled | Metallurgical Silicon Blocks |
This extraction phase provides the raw elemental blocks required for the subsequent complex chemical synthesis.
How is silicon converted into silane precursors?
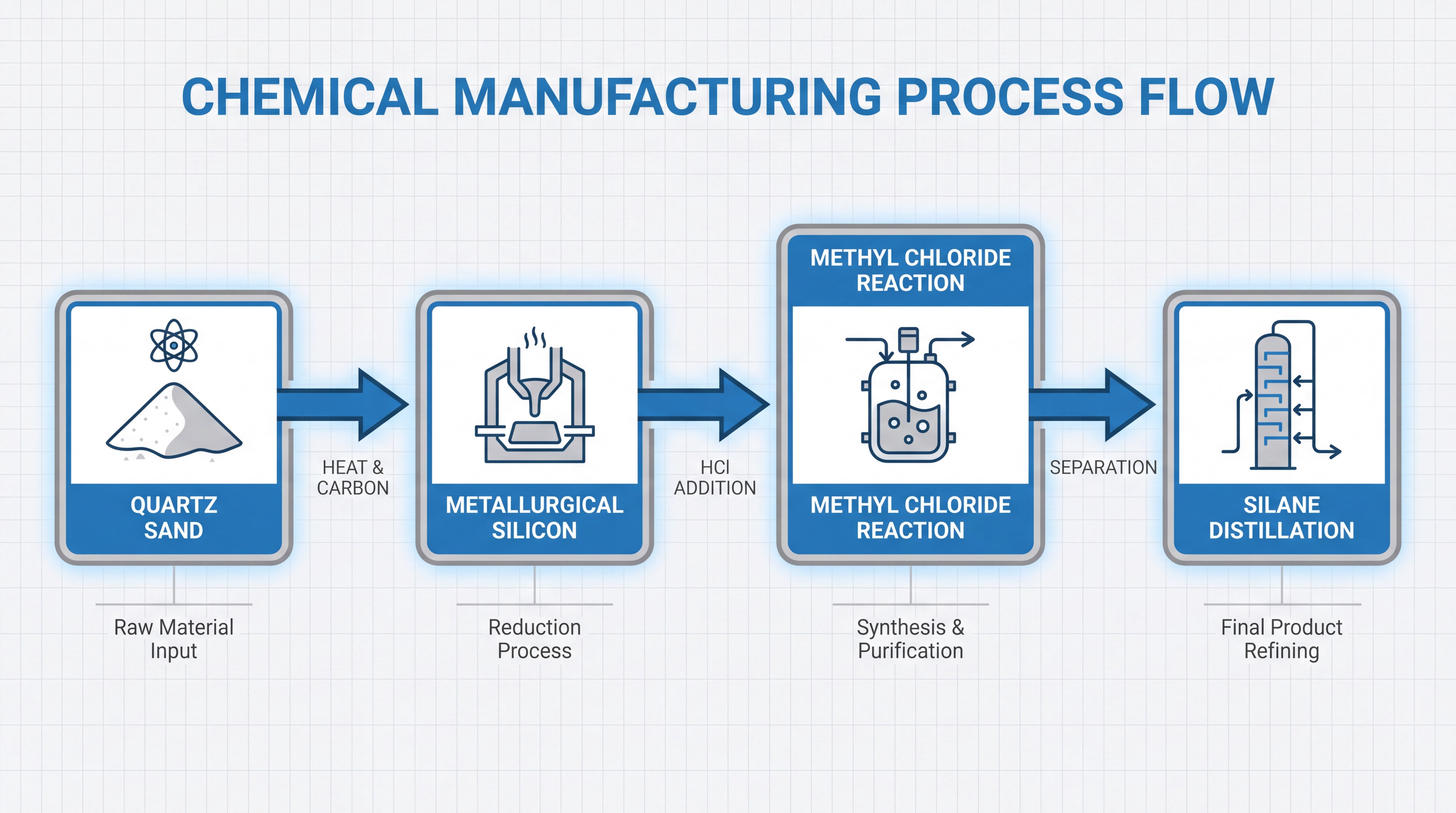
Converted silicon is reacted with methyl chloride in a process known as the “Müller-Rochow” reaction to create various chlorosilanes. This chemical synthesis is a pivotal stage in how to manufacture silicone products because it creates the monomeric building blocks. By reacting fine silicon powder with catalysts, chemists produce dimethyldichlorosilane (DMDCS), which is the most critical precursor for most silicone polymers.
The methyl chloride reaction
The reaction occurs in a fluid-bed reactor where copper catalysts facilitate the bonding of methyl groups to the silicon atoms. Precise control over pressure and temperature is required to maximize the yield of the desired silane types.
Here is the deal:
- The reaction produces a mixture of different silanes that must be separated.
- Distillation columns then isolate the DMDCS from other byproducts.
- This high-purity fluid is highly reactive and serves as the starting point for polymerization.
Key Takeaway: Precision distillation ensures that the precursors are free of impurities that could weaken the final polymer’s molecular structure.
| Component | Role in Process | Chemical Result |
|---|---|---|
| Silicon Powder | Main Reactant | Silicon backbone source |
| Methyl Chloride | Organic Reagent | Adds methyl side groups |
| Copper Catalyst | Accelerator | Drives the reaction efficiency |
Successful silane synthesis allows manufacturers to begin the transition from volatile chemicals to stable, rubbery polymers.
What is the polymerization process for silicone?

Polymerization occurs when silane precursors undergo hydrolysis, which replaces chlorine atoms with hydroxyl groups to form long siloxane chains. When you look at how to manufacture silicone products, this step represents the birth of the actual rubber material. As these chains grow and cross-link, they form a three-dimensional network that gives the material its characteristic elasticity and resilience.
Hydrolysis and chain formation
Adding water to the chlorosilanes triggers a reaction that releases hydrochloric acid as a byproduct while bonding the silicon atoms via oxygen bridges. The resulting mixture contains linear chains and cyclic structures of polydimethylsiloxane (PDMS).
Think about it:
- The length of these polymer chains determines the final viscosity of the silicone oil or gum.
- Short chains produce thin liquids, while long, heavy chains create thick, solid rubbers.
- Condensation reactions further stabilize the molecular weight of the batch.
Key Takeaway: Controlling the polymerization length allows for the creation of everything from thin lubricants to precision wearable parts.
| Feature | Linear Chains | Cross-linked Networks |
|---|---|---|
| Texture | Liquid/Oil | Solid/Rubber |
| Flexibility | High (Flowable) | High (Elastic) |
| Durability | Low | High (Structural) |
This molecular transformation is what allows silicone to maintain its shape after being molded into complex industrial designs.
Which types of silicone exist in the industry?

Industrial silicone is categorized into three main types: Liquid Silicone Rubber (LSR), High Consistency Rubber (HCR), and Room Temperature Vulcanized (RTV) silicone. Navigating how to manufacture silicone products requires choosing the specific variant that fits your production volume and part complexity. Each type offers different processing advantages, from rapid injection molding to manual sealing applications.
Comparing LSR and HCR varieties
Liquid Silicone Rubber (LSR) is a two-component system delivered in a pumpable form, ideal for high-precision, high-volume automated molding. High Consistency Rubber (HCR), also called heat-cured rubber, is a clay-like solid that requires milling and compression molding.
But wait, there’s more:
- LSR provides excellent clarity and is often used for medical components.
- HCR is generally more cost-effective for large, simple parts or extruded profiles.
- RTV silicones are used for potting electronics or on-site sealing where heat curing is impossible.
Key Takeaway: Selecting the right silicone type optimizes your manufacturing speed and ensures the final product meets your specific durability requirements.
| Silicone Type | Consistency | Common Process |
|---|---|---|
| LSR | Pumpable Liquid | Injection Molding |
| HCR | Solid/Clay-like | Compression/Extrusion |
| RTV | Viscous Paste | Casting/Sealing |
Matching the material consistency to the molding machine is the most critical decision in the production planning phase.
How is the raw material compounded for use?

Compounding is the process of mixing the base silicone polymer with reinforcing fillers, catalysts, and pigments to achieve specific physical properties. This is a foundational step in how to manufacture silicone products because raw silicone gum has very little structural strength on its own. By adding silica fillers and chemical agents, manufacturers can customize the hardness, color, and cure speed of the batch.
Integrating fillers and catalysts
Manufacturers use a two-roll mill or a high-shear mixer to fold additives into the silicone base without introducing air bubbles. The most common reinforcing filler is fumed silica, which significantly increases the tensile strength and tear resistance of the elastomer.
Here is the deal:
- Catalysts like platinum or peroxide are added to trigger the curing reaction during molding.
- Pigment masterbatches allow for precise color matching to Pantone or RAL standards.
- Special additives can be included to make the silicone conductive or flame retardant.
Key Takeaway: Custom compounding allows you to engineer a material that perfectly matches your environment, whether it requires extreme heat resistance or medical-grade purity.
| Additive Type | Function | Impact on Part |
|---|---|---|
| Fumed Silica | Reinforcement | Increases tear strength |
| Platinum | Catalyst | Cleaner, faster curing |
| Pigments | Aesthetics | Provides specific color |
Properly compounded material ensures that every molded part performs consistently across thousands of production cycles.
How to manufacture silicone products using molds?

Molding involves placing uncured silicone into a metal cavity and applying heat and pressure to transform it into a permanent solid shape. This is the most visible stage of how to manufacture silicone products, where liquid or solid raw material becomes a functional component. Depending on the design requirements, engineers choose between injection, compression, or transfer molding to achieve the highest precision.
Precision injection and compression
Injection molding is favored for complex LSR parts because it allows for intricate geometries and tight tolerances. Compression molding is more common for solid HCR materials, where a pre-measured “slug” of rubber is pressed into a heated mold.
Think about it:
- Injection molding offers the fastest cycle times for high-volume production runs.
- Overmolding allows silicone to be bonded directly onto plastic or metal inserts.
- The heat in the mold activates the catalyst, causing the polymer chains to cross-link permanently.
Key Takeaway: Choosing the right molding process balances your initial tooling costs against the required part complexity and production speed.
| Molding Type | Material Used | Best For |
|---|---|---|
| Injection | LSR | Complex, high-volume parts |
| Compression | HCR | Large, simple gaskets |
| Extrusion | HCR | Continuous tubing/profiles |
Mastering the molding phase is what allows for the creation of waterproof seals and durable medical valves with micron-level accuracy.
What happens during secondary vulcanization?

Secondary vulcanization, often called post-curing, involves placing finished molded parts into an industrial oven for several hours at high temperatures. This is a crucial step in how to manufacture silicone products meant for sensitive applications like food contact or medical devices. The additional heat ensures that the cross-linking process is 100% complete and that any volatile byproducts are evaporated.
Post-curing for stability
During the primary molding phase, some chemical volatiles may remain trapped within the rubber structure. Post-curing removes these residues, which improves the material’s compression set and eliminates potential odors or “blooming.”
But wait, there’s more:
- It significantly enhances the long-term durability of the silicone in harsh environments.
- Parts are typically baked at 200°C for two to four hours depending on thickness.
- This step is mandatory for medical-grade silicone to meet strict biocompatibility standards.
Key Takeaway: Post-curing provides the final physical stability and safety certification required for high-stakes industrial and healthcare products.
| Property | Before Post-Cure | After Post-Cure |
|---|---|---|
| Volatiles | Present (may outgas) | Removed (inert) |
| Compression Set | Moderate | Excellent (resilient) |
| Odors | Possible | Neutralized |
Stabilizing the polymer through post-curing ensures the parts won’t shrink or change properties after they reach the end user.
How is the quality of silicone rubber tested?

Quality testing involves a battery of physical and chemical assessments to ensure every batch of silicone meets its intended performance specs. When you learn how to manufacture silicone products, you realize that visual inspection is only the first layer of defense. Laboratory technicians perform tensile tests, hardness checks, and compression set evaluations to verify the structural integrity of the components.
Essential physical property tests
Shore A hardness is the most common metric, measured with a Durometer to ensure the part is neither too soft nor too brittle. Tensile testing pulls a sample until it breaks, measuring the force required and the percentage of elongation at the point of failure.
Here is the deal:
- Compression set tests measure how well a gasket recovers after being squashed for a long time.
- For medical parts, extractables testing ensures no harmful chemicals leach out during use.
- Dimensional checks using CCD visual systems verify that tolerances are within specified limits.
Key Takeaway: Rigorous testing protocols prevent field failures and ensure your silicone parts provide a reliable seal or interface for years.
| Test Type | Metric Measured | Why It Matters |
|---|---|---|
| Durometer | Shore A Hardness | Ensures proper tactile feel/seal |
| Tensile | PSI / MPa | Measures structural strength |
| Compression | % Recovery | Crucial for long-term sealing |
Robust quality control turns raw chemical data into a trusted physical product that can withstand extreme industrial stress.
How to choose the right manufacturing partner?

The final step is selecting a partner that offers integrated engineering support, in-house tooling, and stable mass production capabilities. In the journey of how to manufacture silicone products, the factory’s expertise in Design for Manufacturing (DFM) is often the difference between success and failure. A top-tier manufacturer will help you optimize your part geometry to reduce waste and improve the reliability of the finished product.
Evaluating tooling and production
You should look for a partner that manages everything from mold design to final assembly under one roof. This integration prevents “quality drift” that happens when different vendors handle different stages of the process.
Think about it:
- Does the factory have ISO 9001 or medical-grade certifications for your industry?
- Can they provide rapid prototyping to test your design before mass production?
- Do they offer secondary operations like laser marking or adhesive application?
Key Takeaway: The right partner acts as an extension of your engineering team, ensuring that custom silicone solutions are delivered on time and within budget.
| Selection Factor | High-Quality Partner | Low-Quality Vendor |
|---|---|---|
| Engineering | Provides DFM suggestions | Just follows drawings |
| Tooling | In-house mold making | Outsourced (slower) |
| Quality | Traceable materials/ISO | No formal documentation |
Your choice of manufacturer ultimately determines whether your product launches smoothly or faces expensive redesigns later.
Conclusion
Manufacturing silicone is a precise blend of heavy industry and delicate chemistry that requires mastery at every stage. We have explored the entire lifecycle—from the intense heat of silicon extraction to the precision of LSR injection molding and the final stability of post-curing. This article solved the common problem of understanding material differences and provided a clear roadmap for sourcing high-quality elastomers.
We can support your project with in-house tooling, material expertise, and rigorous quality systems. Our vision is to empower global engineers with silicone components that are stable, safe, and perfectly tailored to their unique environments. If you are ready to move from design to reality with a partner that values precision, contact us today for a comprehensive review of your project requirements.
FAQ
Can I use any silicone for food applications?
No, you must ensure the material is specifically labeled as food-grade and complies with FDA or LFGB standards. Standard industrial silicones may contain additives or catalysts that are not safe for ingestion.
What is the best way to bond silicone to plastic?
The best method is overmolding, where the silicone is injected directly onto a pre-molded plastic part. This creates a mechanical or chemical bond that is much stronger than traditional adhesives.
How do I know if I need LSR or HCR?
You should choose LSR if you have high-volume production and complex geometries that require high precision. HCR is generally better for simple gaskets, thick-walled parts, or extruded tubing where cost-efficiency is a priority.
Can silicone parts be recycled?
Yes, silicone can be recycled, though it is usually ground down into “re-grind” and used as a filler for new products. It is not as easily recyclable as common thermoplastics like PET.
How long does a silicone mold typically last?
A high-quality steel mold for injection molding can last for hundreds of thousands of cycles. Aluminum molds are cheaper but will wear out much faster, making them better for prototyping.