Silicone does not melt or burn under standard conditions because it possesses a unique inorganic siloxane backbone consisting of alternating silicon and oxygen atoms. Many engineers and procurement officers worry about the safety of using flexible materials in high-heat industrial or consumer environments. A common concern is: does silicone melt in the oven or release toxic fumes when exposed to a high-temperature baking cycle? Traditional plastics warp, melt, and off-gas at relatively low temperatures, while organic rubbers can catch fire, creating a barrier for those looking to upgrade their hardware. HuaLin Silicone provides high-performance thermosetting elastomers with a unique chemical backbone that remains the gold standard for thermal stability.
What is the chemical secret behind silicone’s heat resistance?
Unlike organic rubbers with a Carbon-Carbon (C-C) backbone, silicone features a robust Silicon-Oxygen (Si-O) chain that resists thermal cleavage. This structural difference is the primary reason engineers ask if does silicone melt in the oven, as the energy required to break a siloxane bond is significantly higher than that of organic polymers. The inorganic nature of the material ensures it retains mechanical integrity when other elastomers would liquefy.
The Strength of the Siloxane Bond?
The siloxane bond is inherently stable and less reactive than carbon bonds found in traditional plastics.
Think about it:
- High bond energy prevents easy thermal breakdown.
- The large atomic radius of silicon allows for greater flexibility without sacrificing strength.
- Inorganic backbones are less susceptible to oxidation.
How Cross-Linking Prevents Liquefaction?
During the curing process, silicone molecules form a three-dimensional network that locks the material into a solid state. This thermoset structure is a key reason why silicone heat insulation pads outperform wood or metal alternatives in thermal management.
| Property | Organic Rubber | Silicone (Siloxane) |
|---|---|---|
| Backbone | Carbon-Carbon | Silicon-Oxygen |
| Bond Strength | ~348 kJ/mol | ~452 kJ/mol |
| Thermal Nature | Thermoplastic/Elastomer | Thermoset Elastomer |
Key Takeaway: The inorganic Si-O bond provides superior thermal stability that prevents melting at temperatures where standard organic materials fail.
The chemical composition dictates that silicone behaves more like a mineral than a typical plastic under thermal stress.
Does silicone melt in the oven during standard baking?
No, standard kitchen ovens rarely exceed 260°C (500°F), which is well below the decomposition threshold of high-quality silicone. When professional chefs ask does silicone melt in the oven, the answer is a definitive no because the material remains solid and functional within common appliance ranges. High-grade silicone is engineered to maintain its shape and elastic properties without leaching chemicals into the environment.
Standard Temperature Ratings for Kitchenware?
Most food-grade silicone products are rated for continuous use up to 230°C.
Here is the kicker:
- Standard baking occurs between 175°C and 200°C.
- Silicone maintains its non-stick properties throughout the cycle.
- Thermal expansion is minimal, ensuring consistent results.
Safety Thresholds for Home Appliances?
Ovens provide convection heat which silicone handles with ease compared to direct conductive heat.
Believe it or not:
- Convection cycles do not reach the 300°C+ required for degradation.
- Silicone mats distribute heat evenly across the surface.
- The material does not off-gas toxic fumes during a standard 400°F roast.
| Appliance Type | Typical Temp Range | Silicone Stability |
|---|---|---|
| Domestic Oven | 150°C – 250°C | Highly Stable |
| Air Fryer | 180°C – 200°C | Highly Stable |
| Microwave | 100°C – 120°C | Completely Safe |
Key Takeaway: Standard kitchen temperatures are insufficient to break the chemical bonds of silicone, making it safe for all common baking tasks.
The safety margin for silicone in domestic appliances is nearly 100 degrees Celsius above common baking requirements.

Why doesn’t silicone ignite like traditional organic rubber?
Silicone possesses a very high ignition temperature, often exceeding 400°C, and exhibits excellent self-extinguishing properties. Even if you are concerned and ask does silicone melt in the oven, you will find that silicone does not support combustion the way wood or plastic does. When exposed to extreme heat, it undergoes a transformation into non-toxic silica rather than fueling a fire.
Self-Extinguishing Properties?
Silicone is often used in fire-stop applications because it does not propagate flames once the heat source is removed.
Think about it:
- The material carbonizes rather than flowing like a liquid fuel.
- It creates a protective “char” layer that insulates the remaining material.
- Low oxygen index ratings make it difficult to ignite in normal atmospheres.
Low Smoke Emission Standards?
Unlike PVC or organic rubber, silicone releases very little smoke when it eventually breaks down.
The bottom line:
- Combustion byproducts are primarily water vapor and carbon dioxide.
- The absence of halogens means no corrosive acid gases are produced.
- It is preferred in aerospace and enclosed transit systems for fire safety.
| Material | Ignition Temperature | Combustion Behavior |
|---|---|---|
| Natural Rubber | ~260°C | Rapid burning, thick smoke |
| Polyethylene | ~340°C | Melting, dripping flames |
| Silicone | >400°C | Self-extinguishing, white ash |
Key Takeaway: Silicone’s high ignition point and lack of flammable dripping make it an ideal material for fire-safe environments.
The material’s transition to silica during combustion acts as a natural fire barrier in many industrial designs.

What happens to silicone at temperatures above 300 degrees?
At temperatures exceeding 300°C, silicone does not turn into a liquid but instead undergoes thermal oxidative degradation. Those wondering does silicone melt in the oven should understand that at these extremes, the polymer chains begin to break and reform into a brittle, white powder known as silicon dioxide. This process leads to a gradual loss of mechanical elasticity and eventual structural failure.
Understanding Thermal Oxidative Degradation?
Oxygen interacts with the silicone chain at high heat, causing the material to harden over time.
Here is the kicker:
- Long-term exposure results in “dry out” and cracking.
- The material becomes less flexible as cross-linking increases.
- Degradation is time-dependent; short bursts of heat are tolerated better.
The Transition to White Ash?
When the degradation is complete, the silicone leaves behind a residue that looks like sand or ash.
Think about it:
- This ash is amorphous silica (SiO2).
- The absence of black soot indicates a high-purity inorganic structure.
- This byproduct is non-toxic and environmentally inert.
| Temperature | Duration | Effect on Silicone |
|---|---|---|
| 200°C | Continuous | No measurable change |
| 300°C | Short-term | Slight hardening |
| 450°C | Immediate | Carbonization and ash formation |
Key Takeaway: Silicone fails by becoming brittle and turning to ash rather than melting into a sticky, flammable liquid residue.
This “dry” failure mode is critical for preventing collateral damage in high-heat industrial failures.
How does food-grade silicone differ from industrial-grade?
The main difference lies in the curing process and purity, though both are highly resistant to heat. When asking does silicone melt in the oven, it is important to choose food-grade variants because they are heat-resistant and non-toxic due to specialized curing methods. Platinum-cured silicone, for instance, ensures no volatile organic compounds (VOCs) migrate into food at high temperatures.
Platinum vs. Peroxide Curing?
Industrial silicone often uses peroxide, which can leave behind “blooming” residues or odors when heated.
The reality is:
- Platinum curing is the gold standard for medical and food safety.
- It results in a higher purity product with zero taste or odor transfer.
- Food-grade silicone must pass rigorous FDA or LFGB extraction tests.
Selecting the Right Products for Custom Needs?
Different applications require specific additives to enhance thermal or chemical resistance.
Think about it:
- Industrial gaskets might prioritize oil resistance over food safety.
- Food-grade molds must maintain flexibility through thousands of heat cycles.
- You can browse specialized products to match your specific heat requirements.
| Grade | Curing Agent | Primary Characteristic |
|---|---|---|
| Industrial | Peroxide | Cost-effective, durable |
| Food-Grade | Platinum | High purity, no odor |
| Medical | Platinum/LSR | Biocompatible, autoclavable |
Key Takeaway: Food-grade silicone is specifically processed to remain inert and safe at high temperatures, unlike some industrial variants.
Ensuring the correct grade is used prevents chemical migration during high-heat cooking or sterilization processes.

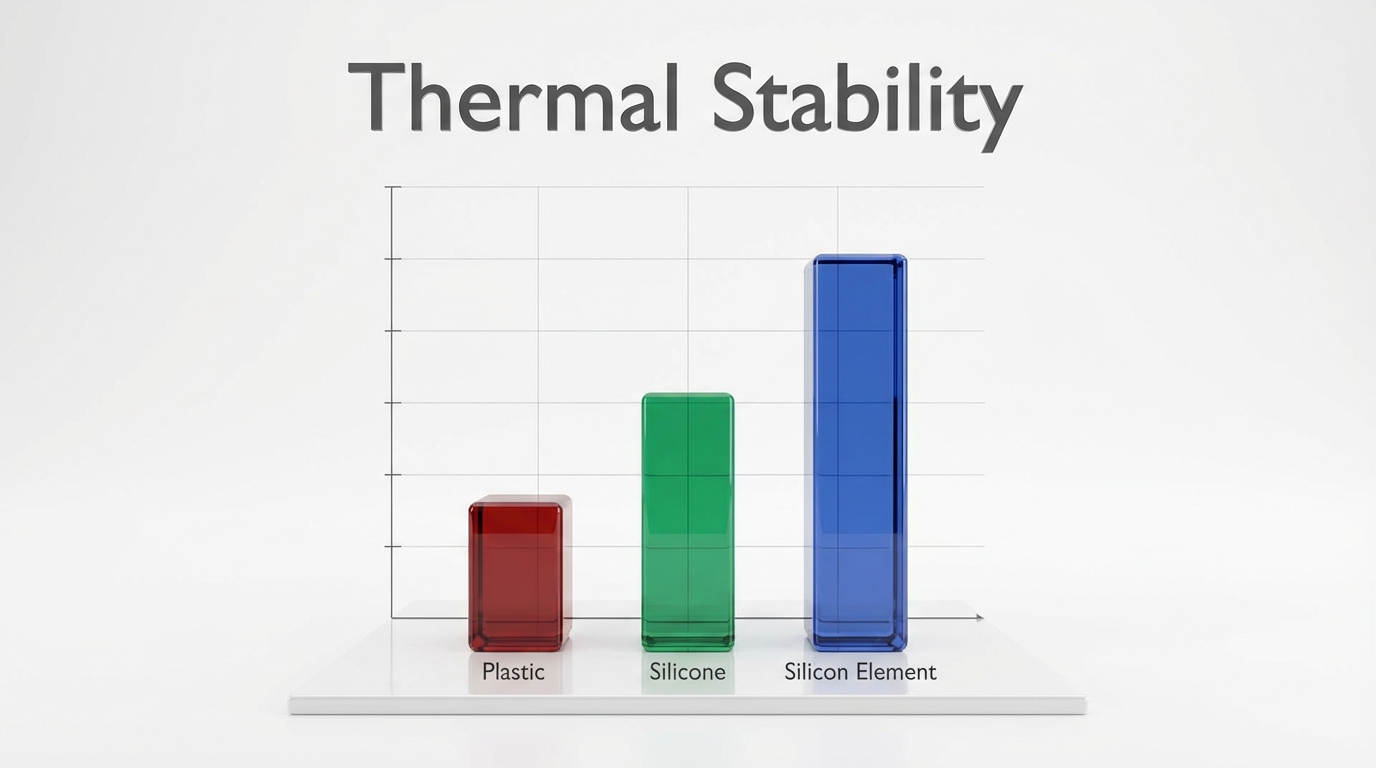
Is there a “melting point” for silicon vs. silicone?
There is a major distinction between the two: Silicon (the element) melts at 1414°C, while silicone (the polymer) has no true melting point. If you investigate does silicone melt in the oven, you will find that as a thermoset polymer, silicone remains solid until it chemically decomposes. It does not transition through a liquid phase because its cross-linked molecular chains are permanently bonded.
Silicon the Element vs. Silicone the Polymer?
Many people confuse the metallic element used in chips with the flexible rubber used in kitchens.
Here is the kicker:
- Silicon is a hard, crystalline solid with a sharp melting point.
- Silicone is a synthetic polymer made of silicon, oxygen, carbon, and hydrogen.
- Mixing these up can lead to significant engineering errors.
Why Polymers Don’t Have a Sharp Melting Point?
Unlike ice or metals, polymers are composed of varying chain lengths that soften gradually.
Believe it or not:
- Thermosets like silicone are “frozen” in their solid state after curing.
- They skip the liquid phase entirely during thermal failure.
- Glass transition temperatures define their flexibility, not their state of matter.
| Material | State | Melting/Decomposition Point |
|---|---|---|
| Silicon (Si) | Crystalline Solid | 1414°C (Melts) |
| Silicone Rubber | Elastomer | ~350°C (Decomposes) |
| Plastic (PE) | Thermoplastic | ~120°C (Melts) |
Key Takeaway: Silicone is a thermoset material, meaning it will decompose into silica powder rather than ever reaching a liquid melting point.
Understanding this distinction is vital for engineers designing high-temperature seals and gaskets.

Can silicone withstand extreme cold as well as high heat?
Yes, silicone is uniquely “amphibious” regarding temperature, maintaining flexibility at -60°C and resisting heat simultaneously. While many worry does silicone melt in the oven, its ability to handle deep-freeze environments is equally impressive for industrial and medical applications. This wide operating range makes it superior to traditional rubbers that become brittle and crack in the cold.
Low-Temperature Flexibility Factors?
The siloxane backbone is highly flexible even when thermal energy is low.
The bottom line:
- Silicone does not undergo a sudden “brittle transition” like most plastics.
- It is often used in baby feeding products that go from freezer to microwave.
- Specialized grades can remain functional down to -100°C for aerospace needs.
Thermal Cycling Stability?
Silicone handles the stress of moving between temperature extremes without structural fatigue.
Think about it:
- It resists “thermal shock” which causes glass or ceramic to shatter.
- Expansion and contraction rates are predictable and stable.
- This makes it ideal for engine seals and outdoor electronics.
| Environment | Temperature | Silicone Performance |
|---|---|---|
| Cryogenic | -100°C | Flexible (Special grades) |
| Arctic | -40°C | Fully functional |
| Industrial Heat | +250°C | Fully functional |
Key Takeaway: Silicone’s ability to remain elastic at sub-zero temperatures makes it one of the most versatile elastomers for global supply chains.
The wide operating window allows one material to serve in both cryogenic and high-heat environments.

How can you tell if your silicone is real using heat?
Real silicone turns into white ash when burned, while “fake” silicone containing plastic fillers will produce black smoke and melt. If you are testing to see does silicone melt in the oven, the burn test is the most reliable method to identify high-purity material. Pure silicone will never drip or create a sticky residue, as the inorganic backbone simply cannot liquefy.
The “White Ash” Burn Test?
Holding a flame to a small corner of the product reveals its true composition.
Here is the kicker:
- Real silicone produces a faint, white glow and no open flame.
- The residue left behind should be a fine, white powder.
- Any black soot or acrid chemical smell indicates the presence of cheap fillers.
The Pinch Test for Plastic Fillers?
Stretching or pinching the material can also reveal hidden impurities.
Think about it:
- Pure silicone maintains its color when stretched or twisted.
- If a white line appears through the material, it likely contains plastic extenders.
- Fillers lower the heat resistance and increase the risk of melting.
| Observation | Pure Silicone | Adulterated “Fake” Silicone |
|---|---|---|
| Smoke Color | None / Very Light | Thick Black |
| Residue | White Powdery Ash | Sticky Black Goo |
| Flame | Self-extinguishing | Maintains flame/Drips |
Key Takeaway: The presence of white ash during combustion is the definitive proof of a high-quality siloxane structure.
Performing a simple burn test ensures that your procurement meets the necessary safety and performance standards.

Does silicone melt in the oven under a broiler?
Direct contact with a broiler element or an open flame can cause silicone to carbonize and fail because the temperatures exceed 500°C. While people ask does silicone melt in the oven, they must realize that “heat-resistant” is not the same as “fire-proof.” Silicone tools should always be kept several inches away from direct heating elements to avoid permanent damage.
Convection Heat vs. Direct Flame?
Ovens use hot air to cook, but broilers use intense infrared radiation that can rapidly overheat surfaces.
The reality is:
- Silicone can withstand the ambient air of a 450°F oven.
- The surface of a broiler element can exceed 1000°F.
- Exceeding 350°C leads to rapid oxidative breakdown.
Safety Guidelines for Professional Kitchens?
Proper placement of silicone tools is essential for longevity and safety.
Think about it:
- Never use silicone mats directly on the floor of a gas oven.
- Ensure mats do not touch the side walls of the oven cavity.
- At HuaLin Silicone, we ensure our materials are tested against these rigorous safety standards.
| Heat Source | Peak Temp | Silicone Status |
|---|---|---|
| Baking (Rack) | 230°C | Safe / Recommended |
| Broiling (Top) | 500°C+ | Danger / Will Carbonize |
| Gas Flame | 1000°C+ | Immediate Failure |
Key Takeaway: Silicone is exceptionally resilient to ambient heat but will fail if subjected to the localized intensity of a broiler or flame.
Maintaining a safe distance from heating elements preserves the molecular integrity of the siloxane bonds.

What are the best industrial applications for silicone?
Silicone’s thermal stability makes it indispensable for automotive gaskets, medical autoclaves, and new energy battery seals. Because engineers know does silicone melt in the oven, they trust it for high-stakes environments where reliability is non-negotiable. Modern manufacturing utilizes new processes like Liquid Silicone Rubber (LSR) injection molding to create complex, heat-shielded components.
Automotive Engine Seals and Gaskets?
Modern engines operate at higher temperatures to increase fuel efficiency, requiring superior seals.
Here is the kicker:
- Silicone remains flexible despite constant heat-cool cycles.
- It resists degradation from engine oils and coolant fluids.
- Vibration dampening is a secondary benefit in harsh engine bays.
Medical Sterilization and Autoclave Parts?
Medical tools must be heated to extreme levels to ensure complete sterilization.
The bottom line:
- Silicone handles repeated steam cycles at 134°C without degrading.
- It is biocompatible and does not harbor bacterial growth.
- LSR molding allows for precision seals in life-saving surgical devices.
| Industry | Application | Why Silicone? |
|---|---|---|
| Automotive | Gaskets/Hoses | Thermal cycling resistance |
| Medical | Tubing/Valves | Autoclavable and pure |
| Electronics | Heat Sinks | Thermal conductivity |
Key Takeaway: The unique thermal properties of silicone allow it to solve engineering challenges that organic rubbers simply cannot meet.
From the operating room to the engine block, silicone remains the material of choice for extreme temperature management.

Conclusion
As a specialized manufacturer in Dongguan, HuaLin Silicone leverages advanced Liquid Silicone Rubber (LSR) and compression molding to create products that defy extreme temperatures. Understanding that silicone decomposes rather than melts is key to its safe application in your next project. Whether you are developing medical-grade components or high-durability consumer goods, our team provides the technical expertise to ensure your designs succeed in the most demanding environments.
Ready to start your custom silicone project? Contact us today for a professional DFM review and RFQ.
FAQ
Can I use silicone molds in an air fryer?
- Judgment: Yes.
- Reason: Air fryers typically operate within the 180°C–200°C range, which is well within the safety limits of high-quality food-grade silicone.
Is silicone toxic if it accidentally burns?
- Judgment: No.
- Reason: The primary byproduct of silicone combustion is amorphous silica (sand), which is non-toxic and inert, unlike the harmful dioxins released by burning plastics like PVC.
Can silicone go directly from the freezer to the oven?
- Judgment: Yes.
- Reason: Silicone has exceptional resistance to thermal shock, meaning its molecular structure won’t crack or fail during extreme and rapid temperature shifts.
Is silicone safer than PTFE (Teflon) at high heat?
- Judgment: Generally, yes.
- Reason: Silicone remains stable up to its limit, whereas PTFE can release toxic “Teflon flu” fumes if accidentally overheated above 260°C.
