Medical grade is a specific designation for materials that meet rigorous biocompatibility and safety standards for healthcare applications. You face immense pressure to deliver medical devices that are both safe and compliant, yet sourcing inconsistent materials can lead to product recalls or patient harm. This uncertainty creates a bottleneck in your development cycle and puts your reputation at risk. HuaLin Silicone offers a controlled manufacturing route for medical grade silicone to eliminate these hazards and ensure your products meet the highest safety standards.
What Is the Definition of Medical Grade Silicone?
Medical grade is a classification for materials that have undergone extensive testing to ensure they do not cause adverse reactions in the human body. Choosing the right medical grade silicone is the first step in ensuring your device passes strict clinical audits and functions reliably.
What are the core safety requirements?
The primary requirement for this material is that it must be non-reactive and stable when in contact with living tissue. You need to ensure that the material does not leach harmful chemicals or cause inflammation during use.
Now, consider this:
- The material must be hypoallergenic.
- It must maintain its physical properties over time.
- Chemical stability is non-negotiable for safety.
How is material classification determined?
Classification is determined by the duration and type of contact the material will have with the human body. You will typically see grades divided into limited, prolonged, and permanent contact categories.
The fact of the matter is:
- Surface devices require different testing than implants.
- Duration of contact dictates the level of scrutiny.
Key Takeaway: Understanding the definition helps you select the correct testing tier for your specific medical device application.
| Feature | Requirement | Benefit |
|---|---|---|
| Biocompatibility | High | Patient safety |
| Chemical Inertness | Extreme | Prevents leaching |
| Stability | Long-term | Device longevity |
This data highlights how fundamental safety requirements translate into direct clinical benefits for your end-users.
Why Is Material Purity Essential for Safety?
Purity ensures that no unexpected chemical reactions occur between the medical device and the patient’s biological systems. High-quality medical grade silicone contains no fillers or additives that could migrate out of the material and compromise the integrity of the medical component.

What contaminants must be avoided?
Contaminants like heavy metals or volatile organic compounds can cause severe irritation or toxic responses. You must ensure your supplier uses platinum-cured systems to avoid the acidic byproducts common in cheaper peroxide-cured silicones.
Here is the truth:
- Platinum curing results in the highest purity levels.
- Residual catalysts must be kept to an absolute minimum.
- Particulate contamination can ruin a cleanroom batch.
How does purity impact patient outcomes?
Higher purity levels directly correlate to lower risks of rejection or infection for the patient. You are providing a safer experience by investing in materials that have been processed with extreme care.
Consider this:
- Pure materials reduce the risk of immune responses.
- Consistent purity leads to predictable healing.
Key Takeaway: Prioritizing material purity eliminates the risk of toxic leaching and ensures your device remains safe for long-term use.
| Factor | Pure Silicone | Low-Grade Silicone |
|---|---|---|
| Catalyst Type | Platinum | Peroxide |
| Byproducts | None | Acidic residues |
| Safety Profile | High | Variable/Low |
Choosing high-purity materials is a critical decision that protects both your patients and your brand’s regulatory standing.
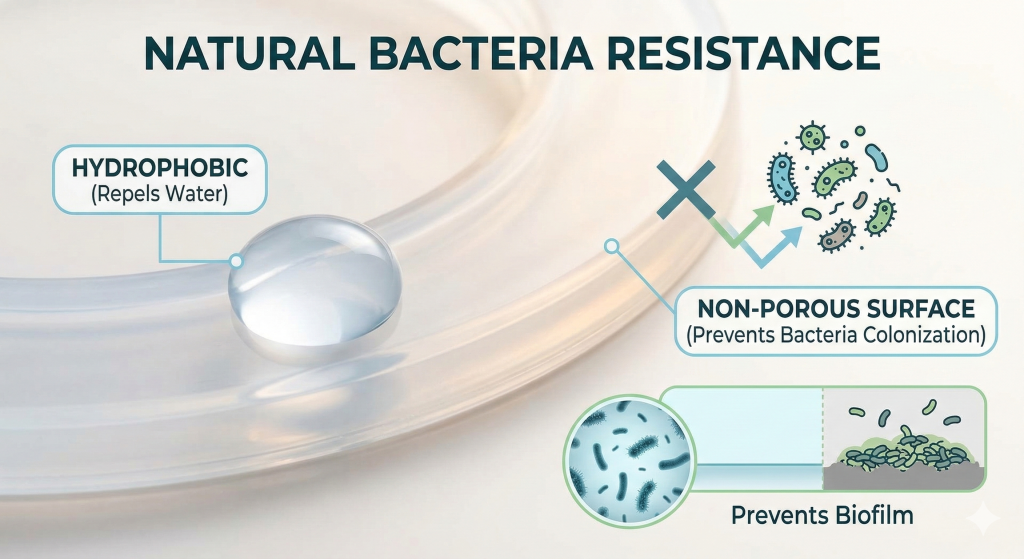
How Does Medical Grade Silicone Resist Bacteria?
Medical grade silicone is naturally hydrophobic and has a smooth surface that makes it difficult for bacteria to colonize. Utilizing medical grade silicone for your seals and gaskets provides an inherent layer of protection against microbial growth in clinical environments.
Why is surface integrity important?
A smooth, non-porous surface prevents microscopic organisms from finding a foothold on the device. You need a material that can be easily cleaned and does not harbor hidden colonies of pathogens.
Let’s look at the facts:
- Smooth surfaces facilitate easier sterilization.
- Low surface energy repels most fluids.
- Structural integrity prevents cracks where bacteria hide.
Can additives enhance resistance?
In some cases, you can integrate antimicrobial agents directly into the silicone matrix for enhanced protection. This creates a secondary defense mechanism for devices used in high-risk infection areas.
Keep this in mind:
- Silver-ion additives are common for antimicrobial needs.
- Additives must not compromise the base material’s safety.
Key Takeaway: The natural resistance of silicone to bacteria simplifies your cleaning protocols and reduces the risk of hospital-acquired infections.
| Surface Trait | Impact | Result |
|---|---|---|
| Hydrophobic | Repels water | Discourages biofilm |
| Non-porous | Smooth finish | Easy to disinfect |
| Chemical Inertness | No nutrients | Bacteria cannot feed |
The combination of physical and chemical properties makes this material an ideal choice for maintaining sterile healthcare environments.
What Standards Govern Medical Grade Materials?
Standards provide a universal framework for measuring the safety and performance of materials used in the medical field. Medical grade silicone must comply with specific testing protocols to be legally sold for use in medical devices.
What is USP Class VI?
USP Class VI is one of the most common certifications, involving a series of in vivo tests to check for toxicity. You should always verify that your material has passed these tests to ensure it won’t cause harm to living tissue.
Here is the reality:
- Class VI involves systemic toxicity testing.
- It includes intracutaneous and muscle implantation tests.
- It is the gold standard for medical plastics.
How does ISO 10993 apply to you?
ISO 10993 is a broader international standard that covers biological evaluation through a risk management process. You must follow this standard to market your device globally and ensure it meets international safety expectations.
Now, consider this:
- It covers cytotoxicity and sensitization.
- It assesses irritation and systemic toxicity.
Key Takeaway: Compliance with USP Class VI and ISO 10993 is essential for any medical device seeking regulatory approval.
| Standard | Focus Area | Application |
|---|---|---|
| USP Class VI | In vivo toxicity | Material validation |
| ISO 10993 | Biological risk | Device certification |
| FDA Registration | Facility audit | Supply chain trust |
Adhering to these standards ensures your products are ready for the global market while maintaining peak safety.
Which Medical Grade Silicone Types Are Available?
Different types of silicone offer varying processing advantages depending on the complexity of your part design. Selecting the right medical grade silicone type allows you to optimize production speed while maintaining tight tolerances for critical components.
What is Liquid Silicone Rubber?
Liquid Silicone Rubber (LSR) is ideal for high-volume, precision injection molding of complex parts. You will find that LSR offers excellent clarity and consistency, which is vital for medical applications requiring high accuracy.
The truth is:
- LSR allows for rapid cycle times.
- It is perfect for intricate, small parts.
- Automated processing reduces human error.
How does High Consistency Rubber differ?
High Consistency Rubber (HCR) is a clay-like material used for extrusion and compression molding. You should choose HCR when you need parts with high tear strength or for specific extruded profiles like tubing.
Think about this:
- HCR is often more cost-effective for large parts.
- It provides superior mechanical durability.
Key Takeaway: Choosing between LSR and HCR depends on your part geometry, volume requirements, and the specific mechanical properties you need.
| Material Type | Process Method | Best For |
|---|---|---|
| LSR | Injection Molding | High-precision/High-volume |
| HCR | Compression/Extrusion | Tubing/Large parts |
| RTV | Room Temp Curing | Seals/Adhesives |
Identifying the correct material type early in the design phase prevents costly tooling changes and production delays.
Why Should You Prioritize Biocompatibility?
Biocompatibility means the material does not produce a toxic or immunological response when exposed to the body. Medical grade silicone is engineered to be biocompatible, making it the safest choice for wearable tech, surgical tools, and implants.
What are the risks of low-grade materials?
Using industrial-grade materials can lead to skin irritation, chemical burns, or chronic inflammation. You risk your entire project’s success if you try to cut costs by using materials that haven’t been certified for medical use.
Here is the truth:
- Industrial silicones contain harmful catalysts.
- Skin contact can trigger allergic reactions.
- Regulatory bodies will reject uncertified parts.
How does biocompatibility aid certification?
Starting with a biocompatible material streamlines your regulatory submission process with the FDA or EU. You can leverage the material’s existing test data to prove the safety of your final device.
Consider this:
- Pre-certified materials save months of testing.
- It builds trust with regulatory auditors.
Key Takeaway: Prioritizing biocompatibility protects your end-users and significantly reduces your path to market by simplifying regulatory hurdles.
| Risk Area | Certified Material | Uncertified Material |
|---|---|---|
| Patient Safety | Guaranteed | Dangerous |
| Regulatory | Fast-track | Likely rejection |
| Brand Image | Professional | High liability |
Investing in biocompatible materials is a strategic move that ensures long-term compliance and user satisfaction for your products.
How Is Medical Grade Silicone Quality Controlled?
Quality control involves rigorous monitoring of the environment, materials, and processes during manufacturing. When producing medical grade silicone, we maintain a strict chain of custody and use precise inspection equipment to catch defects before they leave the factory.

What role does cleanroom molding play?
Cleanrooms prevent airborne particles and oils from contaminating the silicone during the curing process. You need a cleanroom environment to ensure your medical parts are free from debris that could cause device failure.
The bottom line is:
- ISO-certified cleanrooms reduce contamination.
- Controlled humidity prevents material variations.
- Staff must wear specialized protective gear.
How are batch records maintained?
Full traceability is a requirement for medical manufacturing, allowing every part to be tracked back to its raw material batch. You can rely on these records if a quality issue ever arises in the field.
Keep this in mind:
- Every batch has a unique identifier.
- Inspection results are archived for years.
Key Takeaway: Robust quality control and cleanroom environments are essential for producing parts that meet healthcare industry standards.
| QC Step | Tool / Environment | Goal |
|---|---|---|
| IQC | Material Lab | Verify raw purity |
| IPQC | ISO Cleanroom | Prevent contamination |
| FQC | CCD Inspection | 100% visual accuracy |
Tight control over the manufacturing process ensures that every part you receive is consistent with your original specifications.
Can Medical Grade Silicone Be Sterilized?
One of the greatest benefits of medical grade silicone is its ability to withstand repeated sterilization cycles without degrading. Medical grade silicone is compatible with steam, radiation, and chemical sterilization methods, making it highly versatile for reusable devices.

Which sterilization methods are compatible?
Autoclaving (steam) is the most common method, and silicone can survive the high heat and pressure involved. You can also use Gamma radiation or Ethylene Oxide (EtO) without damaging the physical properties of the part.
Here is what you need to know:
- Silicone handles temperatures over 200°C.
- It remains stable during radiation exposure.
- Chemical gas does not penetrate the material surface.
Does sterilization affect part dimensions?
Unlike many plastics, silicone does not warp or shrink during standard sterilization processes. You can trust that your seals and components will maintain their precise fit even after dozens of autoclave cycles.
Consider this:
- Low thermal expansion keeps parts stable.
- The material does not become brittle over time.
Key Takeaway: The ability to endure various sterilization methods makes silicone the ideal choice for reusable surgical instruments and medical equipment.
| Method | Compatibility | Effect on Silicone |
|---|---|---|
| Autoclave | Excellent | No dimensional change |
| Gamma | Good | Slight color shift possible |
| EtO Gas | Excellent | No degradation |
Silicone’s resilience during sterilization ensures that your medical devices can be safely reused, providing better value to healthcare providers.
What Applications Need Medical Grade Silicone?
The versatility of this material allows it to be used in everything from simple tubing to complex implantable components. Using medical grade silicone ensures that these diverse applications remain safe for patients and effective for doctors.
Is it used in surgical instruments?
Many surgical tools use silicone handles for better grip and to withstand the harsh environment of the operating room. You will also see it used in internal components like valves and pumps that handle sensitive bodily fluids.
Now, consider this:
- Ergonomic handles improve surgeon comfort.
- Soft-touch textures provide non-slip grips.
- Resilience is key for mechanical valves.
How do wearable devices utilize it?
Smartwatches and health monitors use silicone straps and sensors because the material is skin-friendly and sweat-resistant. You can create a comfortable, long-wear experience for users by choosing biocompatible grades for these products.
The fact is:
- It doesn’t cause skin rash from sweat.
- It is flexible enough for all-day wear.
Key Takeaway: From the operating room to daily health monitoring, silicone is the material of choice for applications requiring safety and durability.
| Application | Primary Benefit | Key Feature |
|---|---|---|
| Surgical Tools | Sterilizability | High-heat resistance |
| Wearables | Comfort | Skin-safe/Hypoallergenic |
| Fluid Handling | Purity | No chemical leaching |
Understanding these applications helps you see how silicone can solve specific design challenges in your medical product line.
How Do You Choose a Medical Grade Manufacturer?
The right partner should offer more than just molding; they should provide engineering support and a transparent quality system. Medical grade silicone parts require a manufacturer who understands regulatory landscapes and can help you navigate DFM (Design for Manufacturing) challenges.
What certifications should you look for?
You should prioritize manufacturers with ISO 9001 and ISO 13485 certifications, which indicate a focus on medical device quality management. These certifications prove that the factory has the systems in place to manage your project correctly.
Here is the truth:
- ISO 13485 is specific to medical devices.
- FDA registration shows commitment to standards.
- IATF 16949 is a bonus for high-discipline manufacturing.
How do you evaluate factory-direct support?
Direct communication with the engineering team helps you resolve design issues before they become expensive mold errors. You should look for a partner that offers one-stop service from tooling to mass production to reduce supply chain risks.
Consider this:
- In-house tooling speeds up iterations.
- Direct export experience simplifies logistics.
Key Takeaway: Selecting a certified manufacturer with deep engineering expertise ensures your medical project is delivered on time and within specification.
| Criteria | Why It Matters | HuaLin Advantage |
|---|---|---|
| ISO 13485 | Medical compliance | Verified systems |
| In-house Tooling | Lower risk | Faster iterations |
| Export Support | Smooth logistics | Global delivery |
Choosing a partner like HuaLin Silicone provides you with the stability and precision needed for successful medical part production.
This guide has clarified the definition of medical grade, the importance of purity, and the regulatory standards you must meet. We addressed the differences between material types and explained why biocompatibility and sterilization are critical for your success. At HuaLin Silicone, we understand the stakes of medical manufacturing. Our ISO-certified facility and cleanroom capabilities are designed to help you launch safe, compliant products without the typical quality risks of offshore sourcing. We are committed to advancing healthcare through precision silicone engineering and stable mass production.
If you are ready to ensure your next medical project meets the highest industry standards, contact us today for expert DFM advice and a custom quote.
FAQ
Can I use food-grade silicone for a medical device?
No, it is not recommended for medical devices. While food-grade material is safe for contact with edibles, it lacks the specific biocompatibility testing (like ISO 10993) required for medical certification and tissue contact.
How do I know if a material is truly medical grade?
Yes, you can verify this by requesting a Certificate of Analysis (COA) and a material safety data sheet. These documents should show compliance with USP Class VI or ISO 10993 standards and confirm the batch’s testing history.
Is medical grade silicone more expensive than industrial grade?
Yes, it is typically more expensive due to higher purity. The cost reflects the premium raw materials, cleanroom processing requirements, and the extensive safety testing necessary to meet healthcare standards.
Does medical grade silicone ever turn yellow?
No, high-quality medical grade silicone is extremely stable. Unlike some plastics or lower-grade silicones, it is resistant to UV light and oxidation, meaning it will maintain its clarity and physical properties over many years.
Can medical grade silicone be used for long-term implants?
Yes, but only specific “long-term” grades are suitable. You must ensure the material is specifically rated for permanent implantation (over 30 days) and has undergone the most rigorous level of biocompatibility testing.
