Selecting the wrong elastomer for a medical device can lead to catastrophic biocompatibility failures, compromised patient safety, and expensive regulatory delays. When you are caught between the flexibility of TPE and the clinical purity of silicone, the stakes involve more than just “feel”—they involve FDA compliance and long-term durability. This guide on TPE vs. Silicone: Comparing Elastomers for Medical Applications provides a definitive look at these materials to help you scale from prototype to mass production with confidence.
What are the fundamental differences between TPE and silicone?
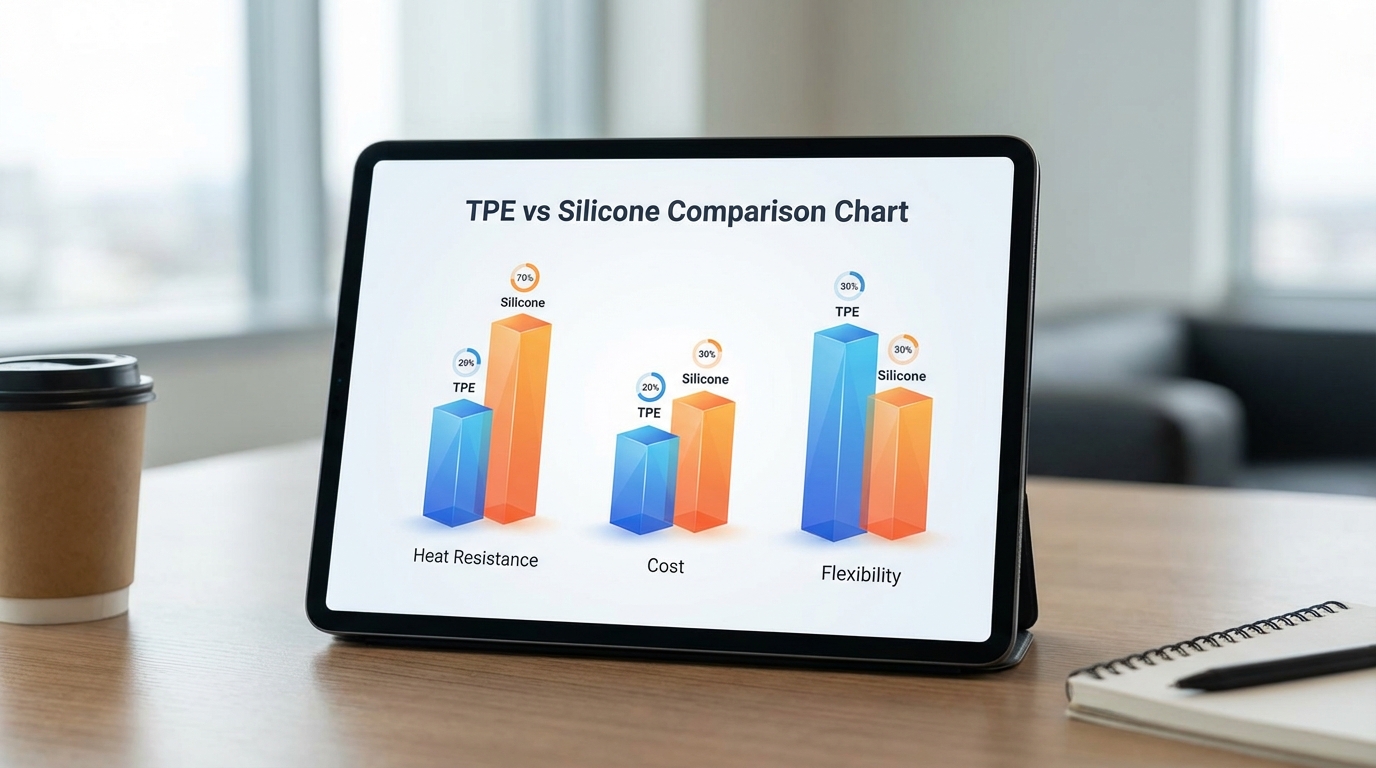
TPE is a thermoplastic that melts when heated, while silicone is a thermoset material that undergoes a permanent chemical cross-linking. Understanding the chemistry behind TPE vs. Silicone: Comparing Elastomers for Medical Applications is essential for any liquid silicone rubber injection molding project. You must decide if your part needs the recyclability of TPE or the structural stability of silicone.
Thermoset Silicone vs. Thermoplastic Elastomers
The primary difference lies in how the polymer chains interact under stress. Silicone uses covalent bonds created during curing to maintain its shape indefinitely.
Think about this:
- Silicone creates a permanent 3D network.
- TPE relies on physical blends that can be re-melted.
- Thermosets offer better “set” resistance over time.
Curing Methods and Material Purity
Medical applications typically require platinum curing to ensure the highest level of purity. This process eliminates the acid residues often found in commercial peroxide-cured materials.
Here is the kicker:
- Platinum curing produces zero harmful byproducts.
- Peroxide curing can leave blooming residues on parts.
- Biocompatibility depends heavily on the chosen catalyst.
Key Takeaway: While TPE is easier to recycle, the cross-linked molecular structure of platinum-cured silicone provides superior purity for critical clinical environments.
| Feature | TPE (Thermoplastic) | Silicone (Thermoset) |
|---|---|---|
| Molecular Structure | Physical Blend | Covalent Cross-links |
| Recyclability | High (Meltable) | Low (Permanent) |
| Chemical Purity | Moderate | Excellent (Platinum-cured) |
The table above illustrates that silicone’s permanent structure is the preferred choice for parts requiring long-term chemical stability.
Why is silicone often the first choice for medical parts?

Silicone consistently leads in high-stakes clinical roles due to its unmatched safety profile and resistance to bacteria. When analyzing TPE vs. Silicone: Comparing Elastomers for Medical Applications, you will find that silicone’s inorganic backbone makes it naturally inert. This quality is vital for devices that remain in contact with the human body for extended periods.
Unmatched Biocompatibility and Purity
Silicone is widely recognized for meeting ISO 10993 standards for skin contact and implantation. Its smooth surface prevents the growth of fungi and bacteria, making it ideal for hospital settings.
Here is why it matters:
- It is non-reactive with human tissue.
- The material does not leach plasticizers.
- It maintains a high level of pharmaceutical-grade purity.
Performance in Clinical Sterilization
Medical devices must survive repeated exposure to heat and chemicals without losing their mechanical properties. Silicone can withstand over 500 alcohol rubs and millions of actuation cycles without cracking.
But wait, there’s more:
- Compatible with Autoclave (steam) sterilization.
- Resists Gamma radiation and ETO gas.
- Retains elasticity after multiple cleaning cycles.
Key Takeaway: Silicone’s inherent resistance to bacteria and harsh sterilization protocols makes it the gold standard for long-term medical device safety.
| Standard | TPE Performance | Silicone Performance |
|---|---|---|
| ISO 10993 | Varies by grade | Consistently Excellent |
| Bacteria Resistance | Moderate | High |
| Sterilization | Limited heat cycles | High-heat compatible |
This comparison highlights why silicone is the safest bet for parts requiring rigorous hospital-grade sterilization.
How does TPE compare in low-volume medical prototyping?

TPE provides a cost-effective and rapid path for disposable medical components and early-stage form-and-fit testing. Understanding how TPE fits into TPE vs. Silicone: Comparing Elastomers for Medical Applications can save you significant time during the initial design phase. It is often the material of choice when you need to iterate quickly before committing to expensive silicone tooling.
The Cost Advantage of TPE Materials
The raw material cost of TPE is generally lower than that of high-performance Liquid Silicone Rubber. For single-use disposable items, TPE offers a budget-friendly alternative that still meets basic safety requirements.
Think about this:
- TPE reduces initial capital expenditure.
- It is ideal for high-volume disposable seals.
- Material waste can be recycled back into production.
Rapid Iteration with 3D Printing
You can use 3D printing technologies like Fused Deposition Modeling (FDM) to create TPE prototypes in hours. This allows your team to test ergonomics and assembly fit without the lead times of traditional molding.
You might be wondering:
- Can I test shore hardness quickly?
- Is it possible to print complex shapes?
- Does it mimic the final silicone feel?
Key Takeaway: TPE offers a faster and more affordable path for prototypes and disposable devices where the extreme performance of silicone is not yet a requirement.
| Prototyping Factor | TPE Approach | Silicone Approach |
|---|---|---|
| Initial Cost | Low | High |
| Speed to Market | Fast (Hours/Days) | Slower (Weeks for tools) |
| Application | Disposables/Testing | High-performance/Implant |
Developers should utilize TPE for early design validation to mitigate financial risks before moving to medical-grade silicone production.
Which material offers better performance in extreme heat?

Silicone remains stable and functional at temperatures that would cause most TPE materials to melt or deform. Thermal stability is often a deal-breaker in medical device design, especially when evaluating TPE vs. Silicone: Comparing Elastomers for Medical Applications. If your device must enter an autoclave or operate near high-heat surgical tools, silicone is the only viable option.
Silicone Operating Range
Silicone thrives in temperatures ranging from -60°C to over +200°C. This massive window allows for use in everything from cryo-storage to high-heat cauterization environments.
Here is the kicker:
- It does not become brittle in the cold.
- It maintains its seal at high temperatures.
- The material properties remain consistent over time.
TPE Thermal Limitations
Most TPE grades begin to soften or lose their structural integrity at temperatures above 100°C. This makes them unsuitable for steam sterilization or high-heat clinical applications.
Think about this:
- TPE is a thermoplastic; it is designed to melt.
- Excessive heat can cause part deformation.
- High-heat sterilization may lead to part failure.
Key Takeaway: For any medical application involving extreme thermal cycles, silicone’s superior heat resistance ensures the device remains functional and safe.
| Temperature Limit | Typical TPE | Medical Silicone |
|---|---|---|
| Low Temperature | -30°C | -60°C |
| High Temperature | +120°C | +200°C+ |
| Autoclave Safe | Usually No | Yes |
Choosing silicone for high-heat environments prevents mechanical failure during critical medical procedures.
Can 3D printing replace traditional silicone molding?

Silicone Additive Manufacturing (SAM) is a breakthrough that allows you to create end-use parts without expensive metal molds. This technology is shifting the traditional view of TPE vs. Silicone: Comparing Elastomers for Medical Applications by offering geometric freedom. While molding is better for mass production, SAM is becoming the new standard for complex, low-volume medical parts.
SAM and the TrueSil Breakthrough
New light-based systems can now cure medical-grade silicone layer by layer. This allows you to build parts with the same mechanical properties as traditional LSR molding.
But wait, there’s more:
- Parts are built from actual silicone, not “silicone-like” resin.
- Resolution is high enough for fine medical features.
- No tooling is required for initial part production.
Geometric Freedom vs. Tooling Deadlocks
3D printing eliminates concerns over flash, voids, and ejection angles that complicate traditional molding. You can design complex lattices and internal channels that are physically impossible to mold.
It gets better:
- You can create custom patient-specific implants.
- Internal structures can be optimized for weight.
- Design changes cost nothing but time.
Key Takeaway: SAM technology allows you to produce true silicone parts with total geometric freedom, bypassing the need for expensive aluminum or steel molds.
| Metric | Injection Molding | SAM 3D Printing |
|---|---|---|
| Geometric Freedom | Moderate | Unlimited |
| Lead Time | Weeks | Days |
| Tooling Cost | High | Zero |
Using 3D printing for complex silicone geometries allows you to move from digital design to physical parts in record time.
How important is biocompatibility when selecting elastomers?
Biocompatibility is the most critical factor for devices like catheters or respiratory tubes that have direct contact with patients. The decision for TPE vs. Silicone: Comparing Elastomers for Medical Applications hinges on the risk of material leaching. While medical-grade TPEs exist, platinum-cured silicone offers the highest assurance of chemical purity.
Pharmaceutical-Grade Purity
Silicone parts often undergo a four-hour post-cure “bake” to remove all volatile organic compounds. This process ensures that the final part is as inert as possible before clinical use.
Here is why it matters:
- It ensures full compliance with ISO 13485.
- It provides batch-to-batch traceability.
- It minimizes the risk of toxic reactions.
TPE Sensitivity and Additives
TPE formulations often require additives to achieve specific levels of flexibility or color. You must be careful to ensure these additives do not leach into the patient’s system over time.
Think about this:
- Certain TPEs may fail USP Class VI tests.
- Some additives are not suitable for implantation.
- Purity levels vary significantly between TPE brands.
Key Takeaway: While TPE is suitable for external medical housings, silicone is the superior choice for any component requiring deep purity and long-term biocompatibility.
| Biocompatibility Factor | Medical TPE | Platinum Silicone |
|---|---|---|
| Leachable Risk | Low to Moderate | Extremely Low |
| Post-Curing Need | Low | High (for purity) |
| FDA Acceptance | Good | Excellent |
The table confirms that silicone’s chemical inertness makes it the safest choice for parts in direct contact with sensitive tissues.
What are the design practices for silicone manufacturability?

Successful silicone design focuses on radii and wall consistency to leverage the material’s unique flow and stretch properties. When designing for TPE vs. Silicone: Comparing Elastomers for Medical Applications, you must follow specific rules to avoid tearing. Silicone behaves differently in a mold than traditional plastics, requiring a specialized approach.
Rounded Corners and Radius Design
Silicone does not like sharp internal edges, which can lead to stress concentrations and tearing during ejection. You should always include generous radii on all internal corners to ensure a clean part release.
Think about this:
- Sharp edges increase the risk of mold damage.
- Rounded corners improve the flow of liquid rubber.
- Consistency in radii leads to fewer part rejects.
Managing Thin Walls and Ejection
The free-flowing nature of silicone allows you to create thin-walled features that would be impossible with other materials. However, you must design these features carefully to prevent “flash” at the parting lines.
But wait, there’s more:
- Silicone’s high elasticity allows for easy “bump-offs.”
- Undercuts can often be ejected without complex slides.
- Tighter tolerances are possible with proper wall planning.
Key Takeaway: Designing for silicone requires a focus on radii and wall uniformity to maximize the benefits of its flow and elasticity.
| DFM Rule | Silicone Requirement | TPE Requirement |
|---|---|---|
| Internal Corners | Must be Rounded | Can be Sharper |
| Undercuts | Easy (due to stretch) | Hard (requires slides) |
| Wall Thickness | High consistency | Flexible limits |
By following these DFM guidelines, you can ensure your silicone parts are both functional and easy to manufacture at scale.
How does overmolding affect choices for medical wearables?

Silicone overmolding is the superior choice for creating waterproof, biocompatible seals on medical wearables. In the debate of TPE vs. Silicone: Comparing Elastomers for Medical Applications, bonding strength is a major factor. Silicone can chemically bond to rigid plastics like polycarbonate, creating an airtight, IP68-rated enclosure.
Silicone-to-Plastic Bonding
Multi-material molding allows you to combine a soft, skin-safe exterior with a rigid structural frame. This 2-shot process is common in high-end smartwatches and medical monitoring devices.
Here is why it matters:
- It creates a permanent chemical bond.
- It provides a seamless waterproof barrier.
- It eliminates the need for messy glues or mechanical seals.
TPE Overmolding Versatility
TPE overmolding is often used for soft-touch grips and shock-absorbing protective covers. While it bonds well to many materials, the bond is often mechanical rather than chemical, which may limit durability.
Think about this:
- TPE is excellent for handheld surgical tools.
- It provides a non-slip grip in wet environments.
- Assembly costs are reduced by molding the grip directly.
Key Takeaway: Silicone overmolding excels in high-purity wearable seals, while TPE overmolding is best for industrial-medical tool grips and protective covers.
| Application | Silicone Overmold | TPE Overmold |
|---|---|---|
| Bonding Type | Chemical | Mechanical/Chemical |
| Water Resistance | IP68 Capable | Basic Sealing |
| Wearable Comfort | Excellent | Moderate |
The table shows that for high-performance wearables, silicone’s superior bonding and comfort are the preferred engineering choices.
Which material provides a better professional surface finish?

Molded silicone offers a smooth, professional clinical finish that is far superior to the granular texture of 3D-printed TPE. Aesthetics and tactile feedback are critical in patient-facing devices, making this a key point in TPE vs. Silicone: Comparing Elastomers for Medical Applications. Silicone’s high resolution and secondary coating options allow for a premium look and feel.
SAM vs. Traditional Molding Surfaces
Parts created through SLS 3D printing often have a “sugar cube” texture that can feel rough to the touch. In contrast, SAM-printed silicone or traditional molded parts have a smooth, layer-free appearance.
Think about this:
- Silicone parts can be made matte or glossy.
- High resolution ensures professional clinical aesthetics.
- Smooth surfaces are easier to clean and disinfect.
Secondary Operations and Texture
You can further enhance the appearance of silicone through laser marking, pad printing, and soft-touch sprays. These coatings are incredibly durable and resistant to the alcohol rubs used in hospitals.
It gets better:
- Laser markings do not fade over time.
- Soft-touch sprays provide a premium “satin” feel.
- Custom colors can be matched to your brand identity.
Key Takeaway: Silicone provides a smoother and more professional aesthetic finish than 3D-printed elastomers, which is vital for patient confidence and device hygiene.
| Aesthetic Feature | 3D Printed TPE | Molded/SAM Silicone |
|---|---|---|
| Surface Texture | Granular/Rough | Smooth/Satin |
| Cleaning Ease | Moderate | Excellent |
| Visual Appeal | Industrial | Professional Clinical |
This data confirms that silicone is the material of choice when surface durability and clinical aesthetics are top priorities.
How do you transition from prototyping to mass production?

Transitioning to mass production requires a long-term view of material stability and detailed DFM reviews from day one. In the context of TPE vs. Silicone: Comparing Elastomers for Medical Applications, you must ensure that your 3D-printed design is actually moldable. Committing to a design that cannot be scaled leads to expensive redesigns and product launch delays.
Avoiding the “3D-Printed Corner”
Designs that work perfectly for SAM 3D printing may have features that are impossible to eject from a traditional steel mold. You must work closely with a manufacturing partner to validate your design for high-volume injection molding.
Here is why it matters:
- It prevents late-stage engineering changes.
- It reduces the cost of secondary operations.
- It ensures part consistency across millions of cycles.
Validating Mass Production Stability
You should implement a three-stage quality control process to ensure every batch meets medical standards. This includes incoming material checks, in-process monitoring, and 100% final inspections.
Think about this:
- IQC verifies the purity of raw silicone.
- IPQC approval is needed for the first article.
- FQC includes dimensional checks and functional tests.
Key Takeaway: Always evaluate your part for mass-production moldability during the prototyping phase to avoid costly redesigned when entering the commercial market.
| Scaling Stage | Prototyping (SAM/TPE) | Mass Production (LSR) |
|---|---|---|
| Tooling Type | Soft/None | Hard Steel |
| Part Unit Cost | High | Low |
| Consistency | Variable | Highly Stable |
Scaling from prototype to production is a complex process that relies on solid DFM foundations and a commitment to clinical-grade quality.
FAQ: Common Questions About TPE and Silicone
- Can I use TPE for permanent medical implants?
Silicone is the primary choice for permanent implants due to its extreme bio-inertness. TPE is generally restricted to transient or external medical applications. - How do I know if my design is moldable for mass production?
You should request a professional DFM review early in the design process to identify potential issues like sharp corners or uneven wall thickness. - What’s the best shore hardness for a medical keypad?
A Shore A range of 40A to 60A is typically best for tactile feedback. This provides the “click” feel users expect while maintaining long-term durability. - Can TPE withstand autoclave sterilization?
Most TPEs cannot withstand the high heat of an autoclave and will deform. Silicone is the preferred choice for devices requiring steam sterilization. - What’s the best way to achieve a waterproof seal on a wearable?
Silicone overmolding on a rigid plastic frame provides the best chemical bond and airtight sealing for IP68-rated medical devices.
Conclusion
This article solved the complex challenge of material selection by comparing mechanical performance, heat resistance, and biocompatibility. Selecting the right elastomer is a balancing act between initial cost, speed to market, and long-term clinical safety. Whether you require the high-purity biocompatibility of platinum-cured silicone for a wearable or the cost-efficiency of TPE for a disposable seal, understanding these material nuances is the key to medical device success. At HuaLin Silicone, we are committed to providing precision-engineered solutions that meet the highest international standards.
Ready to start your next medical project? contact us today for a professional DFM review and custom silicone manufacturing solutions tailored to your requirements.

