Medical Grade Liquid Silicone Rubber is a high-purity, platinum-cured elastomer specifically engineered to meet the stringent safety and performance requirements of the healthcare industry. When you are developing life-critical devices, the risk of material failure or biological incompatibility can lead to catastrophic regulatory delays and patient safety concerns. Standard elastomers often fail to provide the necessary purity, leading to leaching or allergic reactions that compromise your product’s integrity. By utilizing Medical Grade Liquid Silicone, you ensure your components possess the biocompatibility and durability required for both short-term and permanent medical applications.
What defines medical grade liquid silicone rubber?
Medical Grade Liquid Silicone is a thermosetting elastomer characterized by its high purity, chemical inertness, and exceptional thermal stability across a wide temperature range. This material starts as a two-part liquid that is mixed and heat-cured into a solid form, making it ideal for high-precision injection molding.
Core material properties
Medical Grade Liquid Silicone offers a unique combination of flexibility and strength that is unmatched by traditional rubbers.
But that’s not all.
- High transparency for visual monitoring.
- Exceptional dielectric strength for electronics.
- Low compression set for reliable sealing.
- Gas permeability for respiratory use.
Purity and catalyst systems
The use of Medical Grade Liquid Silicone ensures that your device remains free of harmful peroxide byproducts.
Here is why:
- Platinum-curing eliminates acidic residues.
- Minimal volatile organic compounds (VOCs).
- Non-reactive nature with bodily fluids.
Key Takeaway: Understanding the chemical foundation of LSR allows engineers to select materials that prioritize patient safety without sacrificing mechanical performance.
| Property | Medical LSR Value | Benefit |
|---|---|---|
| Hardness Range | 5 to 80 Shore A | Versatile design flexibility |
| Temperature Range | -60°C to +250°C | Withstands all sterilization |
| Elongation | 300% to 1000% | High durability under stress |
This data confirms that LSR provides the necessary physical resilience required for complex medical assemblies.

Why is biocompatibility essential for medical materials?
Biocompatibility is critical because it ensures that Medical Grade Liquid Silicone does not cause toxic, inflammatory, or immune responses when in contact with human tissue. For devices intended for long-term implantation, the material must remain inert to prevent rejection.
Standards for biological safety
Selecting Medical Grade Liquid Silicone involves adhering to global benchmarks like ISO 10993 and USP Class VI.
The best part?
- Rigorous cytotoxicity testing protocols.
- Sensitization and irritation assessments.
- Hemocompatibility for blood-contact devices.
Reducing patient allergic risks
Using Medical Grade Liquid Silicone provides a hypoallergenic alternative to latex and other sensitizing materials.
Believe it or not:
- 100% latex-free composition.
- Resistance to bacterial and fungal growth.
- Soft-touch texture for skin comfort.
Key Takeaway: Biocompatibility certifications are the foundation of trust in medical manufacturing, ensuring every component is safe for human interaction.
| Certification | Focus Area | Application Level |
|---|---|---|
| ISO 10993 | Biological Evaluation | Global standard for all devices |
| USP Class VI | Plastic Safety | High-purity medical requirements |
| FDA 21 CFR | Regulatory Compliance | US market entry necessity |
These certifications serve as a mandatory checklist for engineers during the initial material selection phase.
How does LSR compare to traditional medical elastomers?
Medical Grade Liquid Silicone outperforms alternatives like TPE and HCR by offering superior clarity, lower cycle times, and better environmental resistance. While other resins might be cheaper upfront, they often lack the long-term stability needed for medical environments.
LSR vs. High Consistency Rubber (HCR)
Medical Grade Liquid Silicone is often preferred over HCR due to its automated processing capabilities and reduced waste.
You might be wondering:
- Faster injection speeds reduce costs.
- Higher precision for intricate parts.
- Lower risk of contamination during mixing.
LSR vs. Thermoplastic Elastomers (TPE)
While TPE is easy to process, Medical Grade Liquid Silicone offers significantly better heat resistance and a lower compression set.
The truth is:
- LSR maintains properties after repeated autoclaving.
- TPE can melt or deform at high temperatures.
- Silicone provides better long-term seal integrity.
Key Takeaway: Choosing LSR over cheaper elastomers ensures your product can withstand the rigors of medical sterilization and usage.
| Feature | Liquid Silicone (LSR) | TPE / HCR |
|---|---|---|
| Sterilization | Excellent (Steam/Gamma) | Limited / Moderate |
| Automation | Fully Automated | Often Manual / Semi |
| Precision | High-Tolerance | Standard |
Selecting LSR directly impacts the total cost of ownership by increasing device longevity and reducing failure rates.
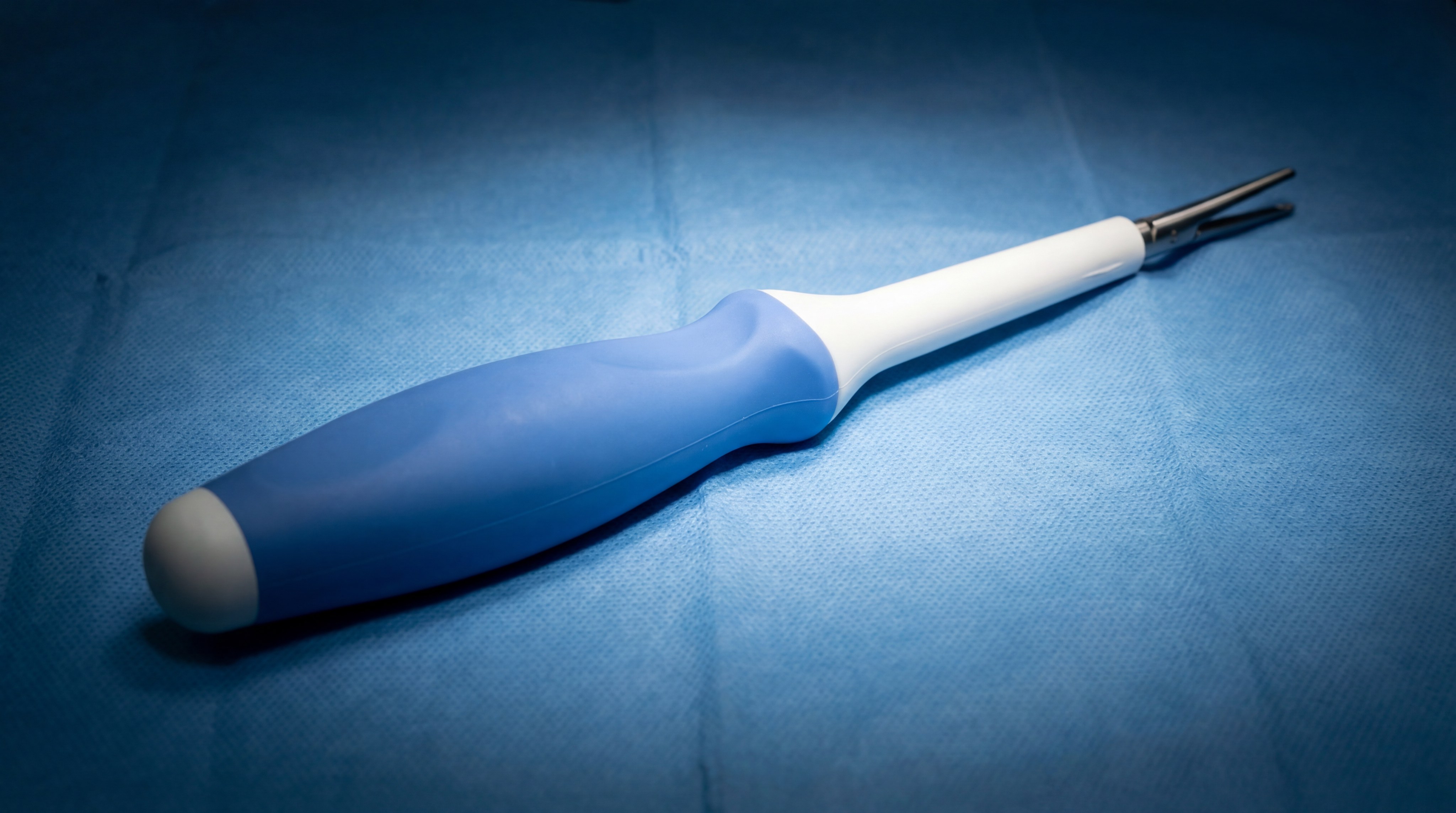
What are the primary medical classifications for silicone?
Medical Grade Liquid Silicone is classified based on the duration and type of contact it has with the human body. These categories—limited, prolonged, and permanent—determine the level of testing and purity required. Using Medical Grade Liquid Silicone ensures you match the right grade to your specific application.
Limited and prolonged contact grades
Medical Grade Liquid Silicone for limited contact is used for devices that touch the skin or mucosal membranes for under 24 hours.
Think about this:
- Used in disposable respiratory masks.
- Ideal for external diagnostic sensors.
- Suitable for temporary wound dressings.
Long-term implantable requirements
Unrestricted Medical Grade Liquid Silicone is required for devices that remain in the body for more than 30 days.
It gets even better:
- High-purity PDMS chemical structures.
- Strict traceability and quality control.
- Used in pacemakers and cardiac stents.
Key Takeaway: Matching the material grade to the contact duration is essential for regulatory approval and patient safety.
| Contact Type | Duration | Example Device |
|---|---|---|
| Limited | < 24 Hours | Syringe seals, masks |
| Prolonged | 24 Hours – 30 Days | Catheters, gastric tubes |
| Permanent | > 30 Days | Orthopedic implants, stents |
Correct classification prevents over-engineering for simple devices while ensuring maximum safety for invasive implants.
Where is LSR used in clinical and surgical settings?
Medical Grade Liquid Silicone is found in a vast array of healthcare products, from anesthesia masks to ergonomic handles for surgical tools. Its versatility allows it to serve both as a primary structural material and a protective coating.
Respiratory and anesthesia care
The use of Medical Grade Liquid Silicone in respiratory equipment ensures a perfect seal and patient comfort.
Here is why:
- High transparency allows fluid monitoring.
- Soft durometer prevents pressure sores.
- Resistant to common anesthetic gases.
Fluid management and catheters
Medical Grade Liquid Silicone is the gold standard for tubes and valves that must transport sensitive fluids without leaching.
But wait, there’s more:
- Smooth surfaces prevent biofilm buildup.
- High tear strength for catheter durability.
- Consistent flow rates in infusion sets.
Key Takeaway: The broad utility of LSR across clinical departments highlights its reliability as a universal medical-grade material.
| Medical Sector | LSR Component | Critical Benefit |
|---|---|---|
| Cardiology | Lead Insulators | Electrical isolation |
| Surgery | Tool Handles | Non-slip, sterile grip |
| Ophthalmology | Contact Lenses | Oxygen permeability |
The integration of LSR into these sectors has directly improved surgical precision and patient recovery times.

How does the injection molding process ensure quality?
The automated liquid injection molding (LIM) process for Medical Grade Liquid Silicone ensures high repeatability and extremely tight tolerances. This method minimizes human error and contamination, which are critical factors in medical manufacturing. Using Medical Grade Liquid Silicone with advanced LIM technology allows for mass production of complex geometries.
Precision and repeatability
Advanced LIM systems for Medical Grade Liquid Silicone provide sub-millimeter accuracy for every part in a high-volume run.
It’s a game-changer:
- Automated mixing of two-part systems.
- Closed-loop control of injection pressure.
- Consistent cure times for uniform quality.
Flash-less and waste-free molding
Medical Grade Liquid Silicone processed via cold runner systems virtually eliminates material waste and secondary trimming.
Consider these facts:
- Reduced risk of particulate contamination.
- Direct-to-package manufacturing potential.
- Lower labor costs for post-processing.
Key Takeaway: Automated molding is the only viable path for producing high-volume medical components that require absolute consistency.
| Process Metric | LIM Standard | Impact on Device |
|---|---|---|
| Cycle Time | 30 – 90 Seconds | High production efficiency |
| Tolerance | +/- 0.05mm | High-precision assembly |
| Waste % | < 2% | Sustainable, cost-effective |
These performance metrics demonstrate why LIM is the preferred manufacturing method for complex silicone medical parts.
Why is overmolding beneficial for medical device design?
Overmolding with Medical Grade Liquid Silicone allows you to bond a soft, protective layer directly onto a rigid plastic or metal substrate. This eliminates the need for adhesives and creates a seamless, monolithic part that is easier to sterilize.
Ergonomic and soft-touch handles
Surgeons prefer tools with Medical Grade Liquid Silicone handles because they provide a secure, non-slip grip even when wet.
Here is the secret:
- Vibration damping for power tools.
- Ergonomic shapes molded to the hand.
- Color-coding for tool identification.
Sterile and bacteria-resistant seals
Overmolding Medical Grade Liquid Silicone creates a permanent bond that prevents liquids and bacteria from being trapped in gaps.
And that’s not all:
- Waterproof seals for electronic devices.
- Seamless transitions between materials.
- Resistance to harsh chemical disinfectants.
Key Takeaway: Overmolding improves both the functionality and hygiene of handheld medical instruments through integrated design.
| Substrate | LSR Bond Type | Common Use Case |
|---|---|---|
| Stainless Steel | Mechanical / Chemical | Surgical scalpels, forceps |
| PEEK / Nylon | Chemical Adhesive | Diagnostic device housings |
| Aluminum | Primer-based | Lightweight medical frames |
The ability to combine disparate materials into a single component reduces assembly time and potential failure points.
What role does a clean room play in LSR production?
A clean room environment is essential for Medical Grade Liquid Silicone production to prevent airborne particulates, dust, and microbes from contaminating the parts. For life-saving devices, even a microscopic contaminant can lead to device failure or patient infection. Using Medical Grade Liquid Silicone in a Class VII or VIII clean room ensures the highest standards of purity.
Contamination control protocols
Clean rooms for Medical Grade Liquid Silicone utilize HEPA filtration and strict gowning procedures to maintain air quality.
The result is clear:
- Prevention of fiber and dust entrapment.
- Reduction in bioburden levels.
- Compliance with FDA and ISO standards.
Ensuring part purity and safety
Processing Medical Grade Liquid Silicone in a controlled environment is mandatory for components like syringe seals and implantable valves.
Here is why it matters:
- Eliminates defects in optical components.
- Ensures consistent seal performance.
- Protects sensitive fluid paths.
Key Takeaway: Clean room manufacturing is not optional for medical devices; it is a fundamental requirement for regulatory compliance.
| Clean Room Class | Max Particles/m³ | Standard Application |
|---|---|---|
| ISO Class 7 | 352,000 | Invasive medical devices |
| ISO Class 8 | 3,520,000 | General healthcare parts |
| Non-Clean Room | Unlimited | Industrial silicone goods |
Maintaining these environmental standards is the only way to guarantee the biological safety of finished medical components.
How can manufacturers optimize costs with liquid silicone?
Cost optimization in Medical Grade Liquid Silicone production is achieved through high-speed automation and integrated part design. While the initial tooling costs for LSR can be higher than other materials, the long-term operational savings are substantial.
Reducing secondary operations
Medical Grade Liquid Silicone allows you to mold complex features that would otherwise require manual assembly or bonding.
Take a look at this:
- Integrated seals reduce part counts.
- Flash-less molding removes trimming labor.
- Automated packaging reduces handling time.
Maximizing production efficiency
The use of Medical Grade Liquid Silicone in multi-cavity molds significantly lowers the per-part cost for high-volume orders.
Here is how:
- Shorter cycle times than compression molding.
- Lower scrap rates through precision LIM.
- High material utilization with cold runners.
Key Takeaway: The total cost of ownership for LSR is lower when considering assembly savings and improved product lifespan.
| Cost Factor | LSR Impact | Long-term Result |
|---|---|---|
| Tooling | High Initial Cost | Durable for 1M+ cycles |
| Labor | Very Low | Automated production |
| Material Waste | Minimal | Efficient ROI |
By focusing on integrated design, manufacturers can leverage the unique properties of LSR to create more value at a lower total cost.
Frequently Asked Questions
Can I use Medical Grade Liquid Silicone for permanent implants?
Yes, specific “unrestricted” grades of Medical Grade Liquid Silicone are tested and approved for permanent implantation exceeding 30 days. These grades undergo the most rigorous biological testing to ensure they remain inert and safe within the human body.
What is the best way to sterilize silicone medical parts?
Absolutely versatile, Medical Grade Liquid Silicone can be sterilized using steam autoclaving, ethylene oxide (ETO), or gamma radiation. Its high thermal stability allows it to withstand repeated sterilization cycles without losing its mechanical properties or flexibility.
How does LSR compare to latex in medical applications?
Medical Grade Liquid Silicone is a superior alternative to latex because it is entirely hypoallergenic and non-toxic. Unlike latex, which can cause severe allergic reactions in many patients, LSR is safe for all skin types and mucosal contact.
Can LSR be overmolded onto plastic medical parts?
Yes, Medical Grade Liquid Silicone is frequently overmolded onto rigid plastics like PEEK or Polycarbonate to create multi-material assemblies. This process often uses self-adhesive silicone grades that bond chemically without the need for additional primers or glues.
What are the common hardness levels for medical silicone?
Medical Grade Liquid Silicone is available in a wide range of durometers, typically from 5 Shore A (very soft) to 80 Shore A (firm). This range allows you to select the perfect balance of softness for patient comfort and rigidity for structural components.
At Hualin, our brand vision is centered on delivering high-precision, biocompatible solutions that solve the most complex engineering challenges in healthcare. We specialize in eliminating the risks of contamination and material failure, ensuring your devices reach the market with the highest safety standards. To discuss your next project or receive a technical consultation on liquid silicone molding, please contact us today.
