Medical grade silicone is a highly specialized, biocompatible elastomer rigorously tested and approved for safe interaction with the human body in healthcare applications. Designing healthcare equipment often brings severe headaches because finding reliable, compliant materials is incredibly difficult. One failed biocompatibility test or minor contamination issue can halt your entire production pipeline, costing you millions in delayed launches and regulatory fines. Here is the deal, choosing the right Medical Grade Silicone solves these problems by providing unparalleled safety, chemical inertness, and proven performance for your most critical projects.
What Is Medical Grade Silicone Exactly?

Medical grade silicone is a premium synthetic polymer explicitly engineered and tested to ensure total biocompatibility with human tissue and bodily fluids. When you manufacture devices for healthcare environments, you cannot rely on standard industrial rubber because it lacks the necessary purity and safety certifications. This highly controlled material undergoes strict validation processes to guarantee it will not cause allergic reactions or release toxic byproducts during use. The manufacturing environment requires absolute cleanliness, often utilizing specialized cleanrooms to prevent any particulate contamination from compromising the final product.
You might be wondering, why is this specific level of purity so heavily regulated by health authorities worldwide? Health organizations demand absolute certainty that materials used in patient care are completely inert and stable under various physiological conditions. The material must maintain its structural integrity without degrading, absorbing fluids, or promoting bacterial growth when placed in clinical settings. By adhering to these stringent standards, manufacturers protect patient health while simultaneously shielding their brands from catastrophic liability issues.
How Does It Perform Under Stress?
Medical grade silicone performs exceptionally well under mechanical stress because of its unique chemical backbone consisting of alternating silicon and oxygen atoms. This strong molecular structure allows the material to flex, stretch, and compress repeatedly without losing its original shape or tearing. This is where it gets interesting…, this resilience is precisely why engineers prefer it for dynamic applications like respiratory masks or pump tubing. You can rely on this material to deliver consistent performance over countless cycles, ensuring medical devices function flawlessly when patients need them most.
- High tear strength ensures longevity during repeated stretching and bending motions.
- Excellent compression set resistance prevents permanent deformation under constant pressure.
- Superior flexibility accommodates dynamic movements required in wearable medical monitors.
Key Takeaway: Utilizing this specialized elastomer guarantees your medical devices will meet rigorous safety standards while delivering reliable, long-lasting performance in critical healthcare scenarios.
How Does It Differ From Standard Silicone?
It differs from standard silicone primarily through its intensive purification process, rigorous biocompatibility testing, and strict regulatory compliance requirements. Standard silicone works perfectly fine for industrial gaskets or automotive seals, but it often contains volatile compounds and trace impurities. If you tried to use industrial rubber in a surgical tool, the chemical leaching could cause severe tissue inflammation or toxic reactions. Therefore, Medical Grade Silicone undergoes specialized curing processes, like platinum curing, to eliminate harmful byproducts and ensure absolute clinical safety.
What is the real story? The difference also extends directly to the manufacturing facilities and the rigorous quality management systems governing production. Facilities producing healthcare materials must operate under strict ISO 13485 standards, maintaining highly controlled cleanroom environments to prevent airborne contamination. Every single batch of material requires exhaustive documentation, creating a traceable history from raw material extraction to the final molded component. This obsessive level of control guarantees that every product reaching a hospital performs predictably and safely.
Why Is The Curing Process Critical?
The curing process dictates the final purity of the silicone, making it a critical factor in differentiating industrial and healthcare-grade materials. Peroxide curing, common in standard manufacturing, leaves behind acidic residues that can alter taste, smell, and biocompatibility profiles. Ready for the good part? Platinum curing systems solve this problem entirely by utilizing a precise catalytic reaction that produces zero volatile byproducts. This advanced technique yields an exceptionally clear, highly pure elastomer that easily passes the most demanding biological reactivity tests.
- Platinum curing eliminates toxic residues that could compromise patient safety.
- The advanced process creates a highly transparent finish ideal for fluid monitoring tubes.
- It significantly improves the overall tensile strength and dimensional stability.
Key Takeaway: Choosing platinum-cured elastomers over standard peroxide-cured options guarantees superior purity, better physical properties, and absolute compliance with medical safety regulations.
| Feature | Standard Silicone | Medical Grade Silicone |
| Curing Method | Often peroxide cured | Strictly platinum cured |
| Biocompatibility | Not tested or verified | Rigorously tested (ISO 10993) |
| Production Environment | Standard factory floor | Certified cleanroom facilities |
| Typical Applications | Automotive seals, cookware | Implants, catheters, tubing |
This table clearly illustrates the stark contrast in quality and compliance between standard elastomers and highly specialized healthcare materials.
What Are The Types Of Medical Grade Silicone?

The primary types include Liquid Silicone Rubber (LSR), High Consistency Rubber (HCR), and Room Temperature Vulcanizing (RTV) silicone. Each specific formulation offers distinct advantages depending on the manufacturing method you choose and the intended clinical application of the device. Liquid Silicone Rubber flows easily into complex molds, making it the perfect choice for high-volume injection molding of intricate healthcare components. Conversely, High Consistency Rubber features a clay-like texture that excels in extrusion processes, typically used for manufacturing long, continuous medical tubing.
Think about it…, selecting the correct type is absolutely vital for optimizing both your production efficiency and the final product’s performance. RTV silicones cure at standard room temperatures, making them highly suitable for creating specialized prosthetics or custom orthopedic impressions without complex machinery. Your engineering team must carefully evaluate the physical demands, production volumes, and sterilization requirements before committing to a specific raw material type. Making the right choice early in the design phase prevents costly manufacturing delays and ensures seamless regulatory approvals.
Which Formulation Suits High-Volume Production?
Liquid Silicone Rubber stands out as the ultimate formulation for high-volume production due to its exceptionally rapid curing times and automated processing capabilities. You can utilize advanced injection molding systems to produce thousands of identical, highly precise components with virtually zero material waste. What’s the catch? The initial tooling costs for LSR injection molds are quite high, requiring a significant upfront capital investment. However, the incredible production speed, consistency, and reduced labor costs quickly offset this initial expense during large-scale manufacturing runs.
- LSR allows for fully automated, continuous production cycles with minimal human intervention.
- It easily accommodates complex geometries, thin walls, and intricate micro-molding details.
- The closed-loop delivery system prevents environmental contamination during the manufacturing process.
Key Takeaway: Investing in Liquid Silicone Rubber and injection molding technology maximizes your production efficiency while maintaining the strict tolerances required for healthcare devices.
Why Do Medical Devices Require This Material?
Medical devices require this material because it provides unparalleled biocompatibility, chemical inertness, and extreme durability under harsh physiological conditions. When you design a device intended for bodily contact, the chosen material must never trigger immune responses or release toxic compounds. Medical Grade Silicone consistently proves itself as the safest option, resisting degradation from bodily fluids, blood, and tissue contact. Furthermore, its inherent flexibility ensures patient comfort, reducing the risk of tissue trauma during prolonged use in sensitive areas.
You might be wondering, how does this material perform when exposed to potent pharmaceutical chemicals or aggressive hospital cleaning agents? The exceptional chemical inertness of this elastomer ensures it will not react with medications, alter drug efficacy, or degrade when sanitized. It withstands exposure to harsh disinfectants without cracking or becoming brittle, ensuring the device remains safe and functional for its intended lifespan. This remarkable stability provides peace of mind for both the healthcare professionals administering care and the patients receiving treatment.
How Does It Improve Patient Comfort?
Patient comfort improves drastically because the material possesses a soft, skin-like tactile feel that minimizes friction and pressure points. If you design a wearable health monitor or a respiratory mask, rigid plastics will inevitably cause severe discomfort and skin irritation. Here is the deal, silicone easily conforms to complex body contours, creating a gentle yet secure seal that patients can tolerate for hours. This superior comfort directly increases patient compliance with therapies, ultimately leading to much better clinical outcomes and improved user satisfaction.
- The soft durometer options prevent pressure ulcers during prolonged skin contact.
- Excellent thermal stability ensures the material does not feel excessively cold upon initial contact.
- Its hypoallergenic nature prevents uncomfortable rashes or allergic contact dermatitis.
Key Takeaway: Utilizing this highly adaptable, comfortable material significantly enhances the patient experience, driving better therapy adherence and superior overall product reviews.
| Material Property | Benefit for Medical Devices | Patient Impact |
| High Biocompatibility | Prevents immune system rejection | Ensures safety during implantation |
| Chemical Inertness | Resists degradation from medications | Guarantees reliable drug delivery |
| Soft Durometer | Conforms to anatomical shapes | Maximizes wearable comfort |
| Hypoallergenic Nature | Avoids triggering skin allergies | Prevents painful skin irritations |
Analyzing these properties reveals exactly why this elastomer dominates the healthcare manufacturing sector and consistently outperforms traditional plastics.
How Is Medical Grade Silicone Manufactured?
It is manufactured in highly controlled, certified cleanroom environments using precision machinery to prevent any microscopic contamination from compromising the batch. The manufacturing journey begins with mixing high-purity raw polymers with specific platinum catalysts to initiate the critical curing process. You must maintain strict temperature and humidity controls throughout the entire facility to ensure the material cures predictably and consistently. Advanced automated systems monitor every single parameter, automatically adjusting pressure and heat to guarantee absolute uniformity across millions of parts.
What is the real story? The post-curing process is equally important, as it involves baking the molded components in industrial ovens to remove any lingering volatiles. This essential step locks in the final physical properties, maximizing tensile strength while ensuring the material meets stringent FDA off-gassing requirements. Quality control inspectors then utilize advanced optical measuring instruments and automated vision systems to meticulously examine each part for microscopic defects. Only after passing these exhaustive dimensional and visual inspections can the components be securely packaged in sterile medical-grade wrapping.
What Role Does Overmolding Play?
Overmolding plays a massive role in modern medical manufacturing by allowing engineers to seamlessly bond soft silicone directly onto rigid plastic substrates. You can create highly complex, multi-material surgical instruments that feature incredibly comfortable, non-slip ergonomic grips designed for precision handling. Ready for the good part? This advanced technique eliminates the need for secondary assembly steps, utilizing mechanical interlocks or chemical bonding to unite the materials permanently. This integration significantly reduces the risk of bacteria accumulating in tiny crevices between assembled parts, greatly enhancing the instrument’s overall hygiene.
- Overmolding drastically reduces assembly time and lowers overall manufacturing labor costs.
- It creates a permanent, watertight seal between rigid housings and soft interactive buttons.
- The seamless transition between materials completely eliminates dangerous bacterial trap zones.
Key Takeaway: Implementing advanced overmolding techniques allows you to design superior, highly ergonomic medical devices while simultaneously streamlining your entire production workflow.
Which Key Properties Make Medical Grade Silicone Stand Out?

Its remarkable thermal stability, exceptional flexibility, and unmatched gas permeability are the key properties that make this material truly stand out. Unlike traditional plastics that melt or shatter under extreme conditions, this robust elastomer maintains its physical integrity across a massive temperature spectrum. When you utilize Medical Grade Silicone, you guarantee your products will perform flawlessly whether they are deep-frozen for storage or steam-sterilized. This broad operational range makes it an incredibly versatile solution for an enormous variety of demanding clinical and laboratory applications.
But here’s the kicker…, the material also exhibits highly unique gas permeability traits that are absolutely crucial for specific medical interventions. For example, certain advanced wound care dressings utilize this permeability to allow oxygen to reach the healing tissue while simultaneously blocking external bacteria. It also prevents moisture buildup beneath the dressing, creating an optimal microenvironment that significantly accelerates the natural cellular regeneration process. These nuanced physical characteristics allow engineers to solve complex physiological challenges that would be impossible with conventional rigid polymers.
Can It Withstand Extreme Temperatures?
It withstands extreme temperatures remarkably well, effortlessly enduring environments ranging from a freezing -60°C up to a scorching 230°C. If you subject standard rubber to an autoclave’s intense pressurized steam, it will rapidly degrade, melt, or permanently deform. This is where it gets interesting…, this specialized elastomer survives thousands of high-temperature sterilization cycles without experiencing any significant loss of tensile strength. This incredible durability ensures expensive reusable surgical tools remain safe and functional for years, providing excellent return on investment for hospitals.
- It survives repeated autoclave cycles without cracking, melting, or shrinking.
- The material remains highly flexible and crack-resistant in deep-freeze storage environments.
- It resists thermal aging, maintaining its original durometer even after prolonged heat exposure.
Key Takeaway: The extraordinary thermal resilience of this material makes it the undisputed champion for reusable medical devices requiring aggressive, high-temperature sterilization protocols.
| Physical Property | Performance Range | Clinical Advantage |
| Temperature Resistance | -60°C to 230°C | Survives aggressive autoclave sterilization |
| Elongation at Break | Up to 1000% | Prevents tearing in dynamic tubing |
| Gas Permeability | Highly permeable to oxygen | Accelerates wound healing processes |
| Hydrophobicity | Repels water naturally | Prevents bacterial adherence on surfaces |
These impressive metrics highlight the precise engineering capabilities that make this elastomer an indispensable asset in modern medical device design.
What Are The Common Medical Devices Made From This Material?
Common medical devices made from this material include intravenous tubing, respiratory masks, surgical drains, and long-term implantable components like pacemakers. You will find this versatile elastomer in almost every hospital department, from the neonatal intensive care unit to the primary surgical suites. Its unique combination of softness and durability makes it ideal for manufacturing delicate infant feeding nipples, ensuring safe and comfortable nutrition delivery. Furthermore, its excellent sealing capabilities are utilized extensively to create waterproof gaskets for expensive, highly sensitive electronic diagnostic equipment.
Think about it…, the sheer diversity of applications clearly demonstrates the incredible trust the medical industry places in this specific polymer. Manufacturers also utilize it heavily in the dental sector to create precise bite blocks, retainers, and highly accurate impression materials. The material’s optical clarity is perfectly suited for creating transparent fluid delivery systems, allowing nurses to visually monitor medication flow rates effortlessly. By choosing this elastomer, you gain access to a material with a proven, decades-long track record of success across countless medical disciplines.
Why Is It Essential For Respiratory Care?
It is absolutely essential for respiratory care because it easily forms a gentle, airtight seal against the patient’s face without causing pressure sores. You must ensure CPAP masks and oxygen delivery systems do not leak, as any lost pressure directly compromises the life-saving therapy. Want to know the best part? The hypoallergenic nature of the material ensures that patients wearing these masks nightly will not develop painful contact dermatitis. It also withstands the constant presence of humidified oxygen without degrading, ensuring the respiratory equipment functions perfectly over long-term usage.
- The soft edges contour precisely to various facial structures for a custom-like fit.
- It prevents localized tissue necrosis during prolonged mechanical ventilation procedures.
- The material easily survives daily cleaning with mild soaps without becoming dangerously brittle.
Key Takeaway: Utilizing this soft, resilient elastomer in respiratory equipment guarantees optimal therapeutic delivery while maximizing crucial patient comfort and long-term compliance.
How Does Biocompatibility Testing Ensure Patient Safety?
Biocompatibility testing ensures patient safety by rigorously evaluating how the material interacts with living tissues, blood, and the human immune system. Before any device reaches a clinical setting, you must submit the raw materials to independent laboratories for exhaustive ISO 10993 evaluations. These strict protocols test for cytotoxicity, ensuring the Medical Grade Silicone does not kill or severely damage surrounding biological cells upon contact. The material also undergoes systemic toxicity testing to guarantee no harmful chemicals leaching into the bloodstream and traveling to vital organs.
Let me explain…, these comprehensive testing protocols are not merely bureaucratic hurdles; they are fundamental safeguards preventing widespread medical disasters. Testing also focuses heavily on sensitization and irritation, verifying that prolonged skin or mucosal contact will not trigger severe allergic reactions. For implantable devices, the material must pass long-term genotoxicity and implantation tests to prove it will not cause tumors or chronic inflammation. Only materials passing every single one of these rigorous biological challenges earn the highly coveted medical-grade classification.
What Is ISO 10993 Certification?
ISO 10993 certification is the internationally recognized standard that provides a structured framework for evaluating the biological safety of medical devices. You must use this specific guideline to determine exactly which biological tests are required based on your device’s intended bodily contact duration. Here is the deal, complying with ISO 10993 is a mandatory requirement for securing FDA approval and successfully launching your product globally. It forces manufacturers to prove scientifically that their chosen materials pose absolutely zero unacceptable risks to the end user’s health.
- It categorizes devices based on contact type: surface, external communicating, or fully implantable.
- It outlines specific testing methodologies for detecting chemical leaching and cellular toxicity.
- It provides a globally harmonized standard, smoothing the regulatory pathway in multiple countries.
Key Takeaway: Achieving ISO 10993 compliance provides undeniable scientific proof that your product is biologically safe, protecting patients and securing your market access.
| Testing Category | Purpose of the Test | ISO 10993 Section |
| Cytotoxicity | Determines if the material kills living cells | ISO 10993-5 |
| Sensitization | Checks for allergic immune system reactions | ISO 10993-10 |
| Systemic Toxicity | Evaluates toxins entering the bloodstream | ISO 10993-11 |
| Implantation | Assesses local tissue reaction over time | ISO 10993-6 |
This testing matrix demonstrates the exhaustive scientific scrutiny required to validate the safety profile of healthcare materials before clinical deployment.
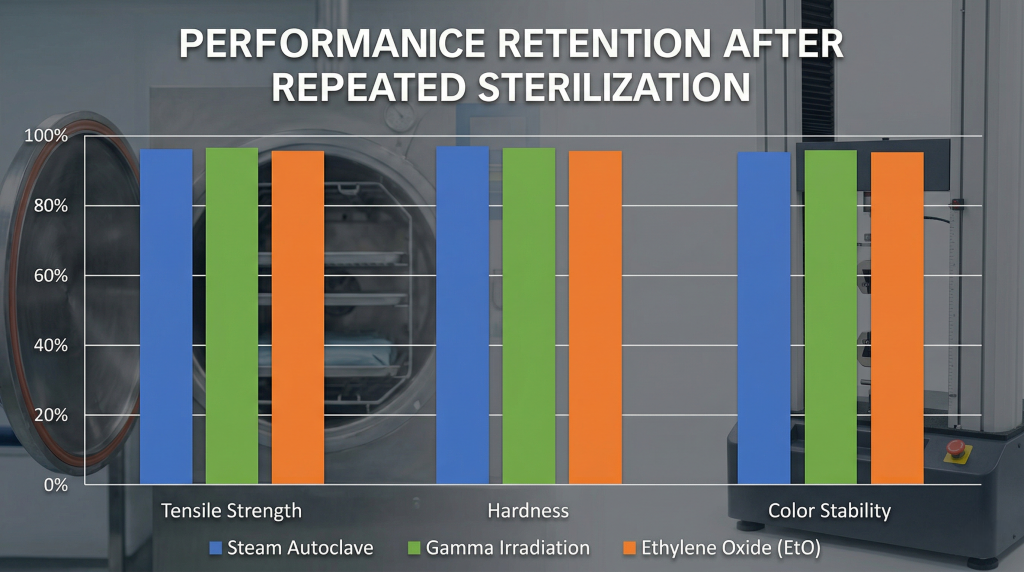
Can It Withstand Harsh Sterilization Methods?
Yes, it can easily withstand harsh sterilization methods, including high-pressure steam autoclaving, ethylene oxide gas exposure, and intense gamma radiation processing. When hospitals reuse surgical tools, you must ensure the materials can survive aggressive decontamination protocols designed to eradicate highly resistant superbugs. Standard plastics warp, melt, or become dangerously brittle when exposed to these extreme sterilization environments, rendering them useless after a single cycle. However, this premium elastomer retains its original mechanical properties, dimensional stability, and functional integrity through hundreds of rigorous sterilization cycles.
You might be wondering, how does the choice of sterilization method impact the long-term performance of the silicone component? While the material excels in autoclaves, it also handles Gamma and E-beam radiation exceptionally well, making it perfect for single-use pre-packaged devices. Ethylene oxide (EtO) gas sterilization is highly effective for complex assemblies, as the permeable nature of the elastomer allows the gas to penetrate deeply. Understanding the interaction between your chosen sterilization protocol and the material ensures you design a highly reliable, compliant healthcare product.
Why Are Reusable Devices Cost-Effective?
Reusable devices are highly cost-effective because the incredible durability of the material allows hospitals to drastically reduce their ongoing supply chain expenses. You can design premium surgical instruments that withstand years of daily use, offering healthcare facilities a significantly better return on their initial investment. Ready for the good part? By eliminating the need to constantly purchase, store, and dispose of single-use plastics, hospitals also massively reduce their environmental footprint. This combination of long-term financial savings and improved ecological sustainability makes durable, reusable equipment highly attractive to modern hospital administrators.
- Durable components withstand thousands of autoclave cycles without requiring costly replacements.
- Reusable designs drastically lower the volume of hazardous bio-medical waste generated daily.
- High-quality, long-lasting tools enhance the perceived value and reputation of your medical brand.
Key Takeaway: Leveraging the immense durability of this elastomer allows you to create sustainable, highly profitable reusable devices that modern healthcare facilities desperately want.
How Should You Choose The Right Medical Grade Silicone Supplier?
Choosing the right supplier requires verifying their quality management certifications, evaluating their cleanroom capabilities, and analyzing their specific industry experience. You must partner with a manufacturer who strictly adheres to ISO 13485 standards, proving they understand the rigorous demands of medical device production. A reliable partner will gladly provide comprehensive material traceability reports, DFM (Design for Manufacturability) support, and robust quality control documentation for every batch. Furthermore, examining their equipment, such as their precision injection molding machines and automated inspection systems, guarantees they can meet your tight tolerances.
What is the real story? A supplier’s ability to communicate transparently and solve complex engineering challenges is just as important as their technical equipment. You need a highly collaborative manufacturing partner who proactively identifies potential design flaws and suggests effective solutions before expensive tooling is cut. Evaluating their past performance with similar medical projects gives you critical insight into their reliability, production speed, and overall commitment to excellence. By carefully vetting your manufacturing partner, you secure your supply chain and drastically reduce the risk of disastrous product recalls.
Why Are Cleanroom Facilities Non-Negotiable?
Cleanroom facilities are absolutely non-negotiable because microscopic dust, airborne particulates, or foreign fibers can critically compromise the safety of a medical device. If you mold a transparent catheter in a standard factory, invisible contaminants can embed themselves in the material, leading to catastrophic patient infections. This is where it gets interesting…, certified ISO Class 7 or Class 8 cleanrooms utilize powerful HEPA filtration systems to constantly scrub the air. This highly controlled environment guarantees the final product remains entirely pristine, easily passing all visual inspections and stringent regulatory cleanliness standards.
- HEPA filters remove 99.9% of dangerous airborne particles from the manufacturing environment.
- Strict gowning protocols prevent human hair and skin cells from contaminating the production line.
- Controlled environments maintain stable humidity, ensuring the elastomer cures perfectly every time.
Key Takeaway: Partnering exclusively with suppliers operating certified cleanrooms is the only way to guarantee your healthcare products remain hygienic, safe, and legally compliant.
| Supplier Criteria | Why It Matters | Red Flag to Avoid |
| ISO 13485 Certification | Proves medical manufacturing competence | Only holds standard ISO 9001 |
| Cleanroom Class | Prevents particulate contamination | Molding happens on open factory floors |
| Material Traceability | Essential for FDA auditing | Cannot track raw material origins |
| DFM Support | Reduces tooling errors and costs | Refuses to review CAD designs |
Evaluating potential partners against this specific criteria matrix significantly reduces your manufacturing risks and ensures a highly successful product launch.
Conclusion
Medical grade silicone has proven itself as the ultimate material for safe, durable, and highly compliant healthcare manufacturing. By understanding its unique properties, rigorous testing requirements, and advanced manufacturing processes, you can design vastly superior products that healthcare professionals trust. We specialize in resolving your complex engineering challenges by providing pristine cleanroom manufacturing and uncompromising quality control tailored to your exact specifications. If you are ready to elevate your next project with precision-engineered elastomers, contact us today to discover how our dedicated team brings your innovative medical visions to life.
FAQ
Q1: Can I use standard industrial silicone for medical devices if I clean it thoroughly?
Absolutely not, because standard silicone lacks the necessary biocompatibility testing and may contain toxic peroxide curing residues. No amount of surface cleaning can remove internal chemical impurities that might leach into the human body and cause severe inflammatory reactions.
Q2: What’s the best way to sterilize products made from this specific elastomer?
High-pressure steam autoclaving is generally the best and most common method because the material easily handles temperatures up to 230°C. However, it also responds exceptionally well to Ethylene Oxide (EtO) gas and Gamma radiation, depending on your device’s specific packaging requirements.
Q3: How do I know if the material I am purchasing is genuinely medical grade?
You must request formal documentation proving the material has passed comprehensive ISO 10993 biological reactivity tests. A reputable supplier will always provide a detailed Certificate of Analysis (COA) explicitly stating the material’s purity and verified biocompatibility profile.
Q4: Can I use this material to create highly complex, intricate shapes?
Yes, utilizing Liquid Silicone Rubber (LSR) injection molding allows you to achieve incredibly precise, complex geometries and micro-features. The low viscosity of the raw material ensures it flows perfectly into the smallest mold cavities before curing into a highly durable solid.
Q5: What’s the best approach to ensuring my manufacturer maintains high quality?
You must exclusively partner with manufacturers who operate certified cleanrooms and hold a valid ISO 13485 medical quality management certification. This guarantees they utilize rigorous, highly traceable production protocols designed specifically to prevent contamination and ensure absolute product consistency.