The primary difference between medical grade silicone and regular silicone lies in their level of purity, the specific curing agents used, and the rigorous biocompatibility testing required for clinical use. Choosing the wrong material for a sensitive application can lead to regulatory failure, skin irritation, or even systemic toxicity for the end-user. Fortunately, utilizing certified medical grade silicone provides the safety profile necessary to protect your reputation and your customers.
What is medical grade silicone exactly?
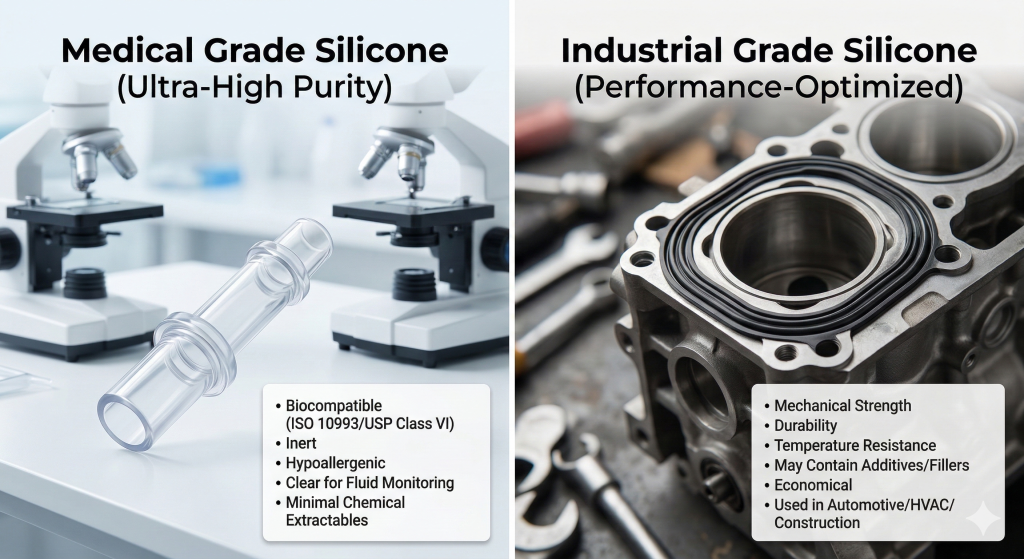
Medical grade silicone is a high-purity elastomer specifically tested for biocompatibility and safe contact with human tissue. This material is designed to be inert and hypoallergenic, making it ideal for the most sensitive healthcare environments. It stands apart from industrial versions due to its lack of toxic fillers and specialized curing processes.
Defining High-Purity Elastomers
Medical grade silicone refers to elastomers that have been specially refined to remove volatile organic compounds and harmful fillers. These materials are formulated to remain inert when in contact with blood, skin, or mucous membranes.
Here is the deal:
- High transparency levels
- Odorless and tasteless
- Minimal chemical extractables
- High thermal stability
Strict Regulatory Oversight
Regulatory bodies demand extensive documentation to prove that a material is safe for human exposure. This involves testing for cytotoxicity, sensitization, and irritation to ensure the material performs consistently under medical conditions. Most manufacturers operate under strict quality management systems to prevent any environmental contamination during the production process.
| Attribute | Medical Grade Standard |
|---|---|
| Purity Level | Ultra-high; no toxic fillers |
| Testing | Biocompatible (ISO 10993/USP) |
| Surface Finish | Smooth, non-porous, and inert |
The specialized processing of medical grade materials eliminates the risk of chemical leaching during use.
Key Takeaway: Medical grade silicone is the only viable option when your product requires absolute safety and documented biological compatibility.
What defines regular or industrial silicone?
Regular silicone is a robust elastomer designed for mechanical performance and environmental resistance rather than biological safety. While medical grade silicone follows strict purity rules, industrial grades prioritize cost-efficiency and durability in harsh settings. These materials are perfect for gaskets, seals, and automotive components where human tissue contact is not a factor.
Performance Over Biocompatibility
Industrial silicone is engineered to withstand extreme temperatures, UV exposure, and mechanical stress. It often contains specific additives that enhance tear strength or flame retardancy, which are beneficial for machines but potentially harmful to humans.
Consider this:
- Optimized for tensile strength
- Formulated for low-cost mass production
- Resistant to oil and environmental weathering
- Wide range of durometers available
Common Additives and Fillers
To achieve specific mechanical properties, regular silicone may include reinforcing fillers like silica or various chemical pigments. While these additives are safe for a car engine or a window seal, they can cause adverse reactions if they come into contact with sensitive biological systems.
| Attribute | Regular Silicone Standard |
|---|---|
| Optimization | Durability and cost-efficiency |
| Additives | Fillers, pigments, and process aids |
| Common Use | Automotive, HVAC, and construction |
Industrial silicone serves as a high-performance mechanical component but lacks the purity required for medical or healthcare sectors.
Key Takeaway: Using regular silicone is a cost-effective strategy for industrial goods where human safety certifications are not a prerequisite.
Is platinum curing better than peroxide?
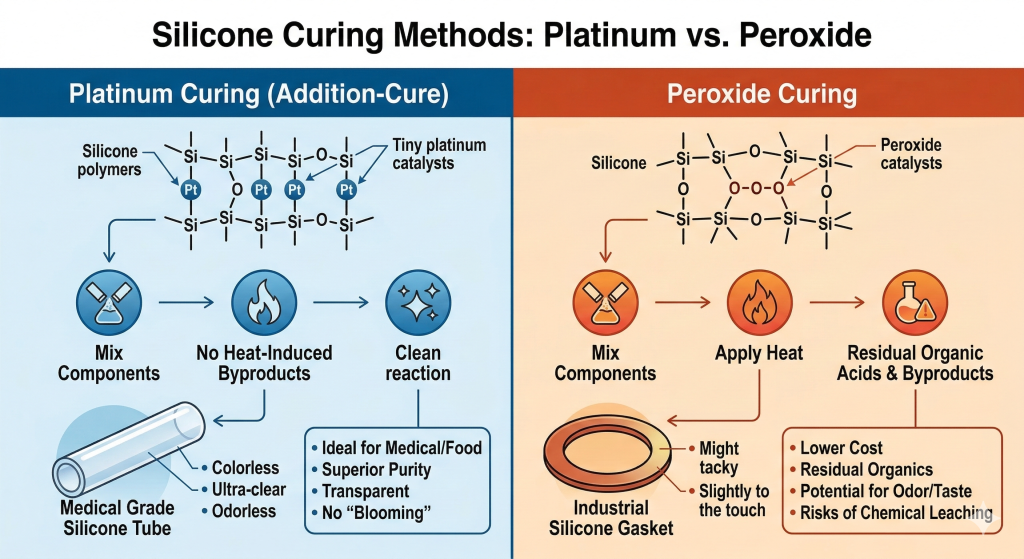
Platinum curing is generally considered superior to peroxide curing for any application requiring high purity and stability. This process uses a platinum catalyst to link silicone polymers without generating any volatile byproducts or organic acids. Consequently, the resulting material is cleaner, more transparent, and less likely to yellow over time.
Cleanliness of Platinum Systems
The addition-cure method involving platinum is the standard for medical and food-grade applications because it is a “clean” reaction. Once the curing is complete, there are no residues left in the material that could leach out into the environment or the patient.
But wait, there is more:
- No “blooming” or white powder residue
- Consistent dimensions after curing
- Superior clarity for fluid monitoring
- Higher tear strength in many cases
Residuals in Peroxide Curing
Peroxide curing is a traditional method that is often used for industrial silicone due to its lower cost and versatility. However, it leaves behind trace organic acids and byproducts that can cause the material to have a slight odor or taste. These residues can also migrate to the surface, which is why peroxide-cured parts are rarely suitable for internal medical use.
| Curing Method | Key Characteristic | Application Suitability |
|---|---|---|
| Platinum Cure | No byproducts, ultra-stable | Medical, Baby, Food |
| Peroxide Cure | Residual organic acids | Industrial, Automotive |
Platinum curing processes ensure the final product remains chemically inert and safe for sensitive user interactions.
Key Takeaway: Choosing platinum-cured silicone is an investment in product purity and long-term material stability.
Which certifications prove medical safety?
Certifications like USP Class VI and ISO 10993 serve as the definitive proof that a material is truly medical grade. When you specify medical grade silicone, you are choosing a material that has undergone rigorous in vivo and in vitro testing. These standards ensure the silicone does not cause toxicity or adverse reactions when exposed to the human body.
USP Class VI Standards
The United States Pharmacopeia (USP) Class VI is one of the most common benchmarks for medical plastics and elastomers. It involves injecting the material into laboratory animals to monitor for any systemic toxicity or local tissue irritation.
Think about it:
- Tests for acute systemic toxicity
- Evaluates intracutaneous reactivity
- Checks for muscle implantation reactions
- Guarantees high material stability
ISO 10993 Compliance
While USP Class VI is a popular standard, ISO 10993 is the international gold standard for biological evaluation of medical devices. This multi-part standard covers everything from surface irritation to long-term chronic toxicity and hemocompatibility.
| Certification | What It Proves |
|---|---|
| USP Class VI | Material is non-toxic to living tissue |
| ISO 10993 | Comprehensive biocompatibility testing |
| FDA 21 CFR | Safe for repeated food contact |
Regulatory certifications provide the objective evidence required to clear medical devices through global health authorities.
Key Takeaway: Never assume a material is medical grade without verifying its USP Class VI or ISO 10993 test reports.
Why is material purity so critical?
Material purity is critical because even trace amounts of contaminants can compromise the safety of a medical device or a sensitive consumer product. High-purity silicone is free from heavy metals, phthalates, and latex, which are common sources of allergic reactions. By maintaining a clean chemical profile, manufacturers ensure that the silicone does not interfere with drug formulations or bodily fluids.
Avoiding Leachable Substances
In a clinical setting, “leachables” are chemicals that migrate from the silicone part into a patient’s bloodstream or medication. High-purity medical silicone is specifically designed to have zero extractables, preventing chemical contamination during use.

The bottom line is:
- Prevents drug-material interactions
- Reduces the risk of inflammation
- Ensures patient safety during long-term use
- Maintains material integrity in fluids
Impact on Transparency
Purity also affects the physical appearance and performance of the silicone, particularly its clarity. Higher purity silicone allows for better visual monitoring of fluids in tubes or masks, which is a vital safety feature for medical professionals. Impure silicone may appear cloudy or yellowed, making it difficult to detect air bubbles or blockages in the system.
| Purity Factor | Medical Impact |
|---|---|
| No Fillers | Prevents allergic skin reactions |
| Zero Extractables | Protects chemical purity of drugs |
| High Clarity | Allows for visual fluid inspection |
Achieving maximum purity requires a controlled manufacturing environment and premium-grade raw materials.
Key Takeaway: Purity is not just an aesthetic choice; it is a fundamental safety requirement that prevents chemical migration.
Where should you apply medical silicone?
Medical grade silicone is the standard choice for any application that involves prolonged contact with the human body or sensitive fluids. For instance, medical grade silicone is essential in infant products to ensure no harmful substances are ingested during use. From surgical implants to respiratory masks, its versatility and safety make it indispensable in the healthcare industry.
Healthcare and Pharma Uses
In the medical field, this material is used for components that must be sterilized and remain inert. This includes catheters, IV tubing, and seals for drug-delivery pumps where chemical stability is non-negotiable.
Look at it this way:
- Surgical tools and grips
- Long-term implants
- Pharmaceutical processing tubes
- Diagnostic equipment seals
Safety in Infant Care
Because infants have developing immune systems, the materials used in their products must be ultra-safe. Medical grade silicone is the preferred material for pacifiers, bottle nipples, and teethers because it can be easily cleaned and contains no BPA or toxins.
| Application | Product Examples |
|---|---|
| Infant Care | Pacifiers, teethers, bottle nipples |
| Medical Devices | Catheters, gaskets, breathing masks |
| Life Sciences | Lab-on-a-chip, peristaltic pump hoses |
Using the correct grade in these applications ensures compliance with global safety regulations and protects vulnerable users.
Key Takeaway: Any product destined for medical use or infant care must prioritize the safety of medical grade materials.
In what cases is regular silicone best?
Regular silicone is the best choice when the primary requirements are mechanical strength, temperature resistance, and cost efficiency. It is the workhorse of the industrial world, providing reliable performance in environments where biological safety is not a concern. For automotive parts or industrial gaskets, medical grade purity would be an unnecessary expense that offers no functional advantage.
Industrial and Mechanical Needs
Industrial grades are formulated to handle high pressure, friction, and extreme thermal cycling. These materials are found in everything from spark plug boots in cars to window seals in high-rise buildings.
Let’s dive deeper:
- Excellent compression set resistance
- High vibration damping capabilities
- Superior resistance to ozone and UV
- Cost-effective for high-volume orders
Cost-Effective Consumer Goods
For many general consumer products, regular silicone provides the perfect balance of durability and price. Items like remote control covers, kitchen spatulas (food-grade), and electronics protective cases do not require medical-level certifications to be effective.
| Industrial Sector | Common Components |
|---|---|
| Automotive | Radiator hoses, gaskets, O-rings |
| Consumer Tech | Keypads, phone cases, cable sleeves |
| Industrial HVAC | Oven seals, boiler gaskets, insulation |
Regular silicone offers the necessary mechanical properties for non-clinical applications at a much lower price point.
Key Takeaway: Use regular silicone for industrial and consumer applications where mechanical performance is more important than biocompatibility.
Can you sterilize regular silicone parts?
While regular silicone is heat resistant, it is not always designed to withstand the repeated, harsh sterilization cycles common in medical environments. Medical grade silicone is specifically engineered to maintain its physical properties after exposure to autoclaving, gamma radiation, or ethylene oxide. If you attempt to sterilize regular silicone repeatedly, it may become brittle, change color, or lose its sealing ability.
Stability Under Extreme Heat
Medical grade silicone maintains its elasticity and structural integrity even after being subjected to high-pressure steam in an autoclave. This allows medical devices to be reused safely multiple times without degrading the material.
It gets even better:
- Retains flexibility after radiation
- Withstands chemical disinfectants
- Resists yellowing from UV light
- Maintains tight tolerances after heating
Degradation Risks
Industrial silicones may contain stabilizers or peroxide residues that react poorly to sterilization processes. Over time, the material may “outgas” or release chemical odors when heated, which could contaminate a sterile environment.
| Method | Medical Grade | Regular Grade |
|---|---|---|
| Autoclave | Highly stable | Risk of hardening |
| Gamma Ray | Very resistant | Potential for yellowing |
| Ethylene Oxide | Safe for use | Variable compatibility |
Specifying medical grade materials ensures that your components will survive the sterilization protocols required for clinical reuse.
Key Takeaway: Medical grade silicone is essential for any part that must be sterilized to prevent bacterial growth and material failure.
How does the cost compare to performance?
Medical grade silicone carries a higher price tag than regular silicone due to the cost of raw materials and cleanroom production. However, when evaluating cost, you must consider the “cost of failure” in your specific application. In a medical or safety-critical environment, the investment in high-quality material is a insurance policy against product recalls and legal liabilities.
Strategic Cost-Benefit Analysis
For a medical device startup, the higher cost of platinum-cured silicone is justified by the speed of regulatory approval. Using a pre-certified material reduces the amount of testing you must perform on the final assembly.
Make no mistake:
- Higher raw material costs (Platinum)
- Strict cleanroom labor expenses
- Documentation and traceability costs
- Reduced risk of product liability
Managing Project Budgets
Smart procurement involves using medical grade silicone only where it is truly necessary—the “wetted” parts of a device. Non-critical housing or external gaskets can often be made from high-quality industrial silicone to balance the overall project budget.
| Factor | Medical Grade | Regular Grade |
|---|---|---|
| Price Point | Premium | Economical |
| Risk Profile | Low (Validated safety) | Higher (If used in medical) |
| QC Requirements | Batch-to-batch traceability | Standard industrial QC |
Choosing the right grade based on functional needs allows you to optimize costs without compromising on essential safety.
Key Takeaway: Medical grade silicone provides a high return on investment through risk mitigation and guaranteed regulatory compliance.
How do you select a silicone manufacturer?
Selecting a manufacturer for medical-grade parts requires a deep dive into their quality management systems and factory certifications. A reputable partner should hold ISO 13485 certification, which is the specific standard for medical device manufacturing. They must also demonstrate full traceability of their raw materials and maintain a clean production environment to prevent cross-contamination.

Evaluating Quality Management
A professional factory will have documented procedures for every step of the molding process. This ensures that every batch of parts produced is identical in quality and purity, which is vital for clinical applications.
Truth be told:
- ISO 13485 is a must for medical parts
- Cleanroom facilities prevent defects
- Material traceability protects your supply chain
- In-house testing ensures compliance
Support for Design
The best manufacturers offer Design for Manufacturing (DFM) support to help you optimize your parts for consistent quality. They can advise on wall thickness, gate placement, and material selection to ensure your medical device performs perfectly from the first prototype to mass production.
| Criteria | Why It Matters |
|---|---|
| ISO 13485 | Ensures medical-level quality control |
| Cleanroom (Class 100k) | Minimizes airborne contamination |
| DFM Expertise | Reduces production errors and costs |
Partnering with an experienced manufacturer ensures that your material choice translates into a high-quality finished product.
Key Takeaway: Your supplier’s quality systems are just as important as the silicone grade itself when manufacturing medical products.
FAQ: Common Questions About Silicone Grades
- Can I use regular silicone for food items?
Regular silicone is not automatically safe for food contact unless it is specifically certified as FDA food-grade or LFGB. While industrial silicone is non-toxic in its solid state, food-grade versions are tested to ensure no chemicals migrate into food at high temperatures. - What’s the best way to identify medical silicone?
Visually, it is nearly impossible to tell the difference, but medical grade silicone is usually more transparent and completely odorless. The only definitive way to identify it is by reviewing the manufacturer’s material certification and batch test reports. - Is medical grade silicone the same as surgical grade?
Surgical grade is a subset of medical grade silicone that is tested for long-term implantation inside the body for more than 30 days. Most medical grade silicones are intended for short-term contact or external use, while surgical grades meet even stricter biocompatibility thresholds. - Why is platinum curing preferred for medical use?
Platinum curing is preferred because it does not produce any volatile byproducts or organic acids during the cross-linking process. This results in a cleaner, more stable material that is free from extractables that could harm patients. - Does medical silicone have a shelf life?
While the silicone material itself is extremely stable, the certifications and sterilized packaging often have expiration dates. Most raw silicone elastomers have a shelf life of 6 to 12 months before they are molded, after which the finished part is stable for many years.
Conclusion
The distinction between medical grade and regular silicone is vital for any business involved in product development, manufacturing, or healthcare. By understanding the differences in purity, curing methods, and certifications, you can ensure your products meet the highest safety standards while optimizing your production costs. Whether you are building life-saving medical devices or durable industrial components, choosing the correct material grade is the foundation of a successful product.
At HuaLin Silicone, we solve complex material challenges by providing high-purity components and expert engineering guidance. We can help you navigate regulatory hurdles and optimize your designs for mass production. If you are ready to start your next project with a partner who understands the importance of precision and safety, contact us today for a comprehensive consultation and professional DFM support.
