Medical Grade Liquid Silicone Rubber (LSR) is a high-purity, platinum-cured elastomer specifically engineered to meet the stringent biocompatibility and safety requirements of the healthcare industry. Imagine a scenario where a critical surgical valve fails mid-operation due to material degradation or leaching contaminants into a patient’s bloodstream. This nightmare for medical manufacturers isn’t just a quality control issue; it is a catastrophic risk to patient life and brand reputation. To eliminate these vulnerabilities, leading engineers utilize medical grade silicone for molding to produce chemically inert, highly durable components that withstand repeated sterilization and maintain peak performance in life-critical environments.
What is Medical Grade Liquid Silicone Rubber (LSR)?

Medical Grade Liquid Silicone Rubber is a specialized, inorganic elastomer composed of a backbone of alternating silicon and oxygen atoms that provides exceptional thermal and chemical stability. Using medical grade silicone for molding allows manufacturers to create parts that are inherently non-toxic and odorless.
Material Composition and Purity
LSR is a two-part system—Part A and Part B—that, when mixed in equal ratios, undergoes a chemical reaction to form a solid, cross-linked polymer. This material is prized for its high purity, as it is cured using a platinum catalyst rather than peroxide.
- Platinum-cured for zero acid byproducts.
- High transparency for visual flow monitoring.
- BPA-free and latex-free composition.
The Thermosetting Advantage
Unlike thermoplastics that can melt when reheated, LSR is a thermosetting material that remains solid once cured. Here is the kicker: this permanent state makes it the ideal choice for medical devices that must undergo high-heat autoclaving without losing their shape or functional integrity.
Key Takeaway
LSR represents the gold standard for medical purity, offering a stable, bio-inert foundation for the world’s most sensitive healthcare applications.
| Feature | Specification |
|---|---|
| Curing Agent | Platinum |
| Purity Level | Medical/Healthcare Grade |
| Physical Form | Liquid Pumpable Paste |
| Post-Curing | Permanent (Thermoset) |
LSR provides the necessary safety margins required for components that have direct or indirect contact with the human body.
Why is Biocompatibility Critical in Medical Silicone?

Biocompatibility is critical because it ensures that the medical grade silicone for molding does not induce toxic, immunological, or carcinogenic responses when in contact with living tissue. Ensuring your material is bio-inert is the only way to safeguard patients during prolonged clinical exposure.
ISO 10993 Compliance Standards
Medical-grade materials must pass rigorous testing protocols to be certified for clinical use. But there is a catch: not all “silicones” meet these standards, making it vital to verify compliance documents from your supplier.
- Sensitization and irritation testing.
- Systematic toxicity evaluations.
- Hemocompatibility for blood-contacting devices.
Risk Mitigation in Patient Care
Using a certified biocompatible material significantly reduces the risk of inflammation or rejection in the patient. This predictability is what allows surgeons to trust silicone-based implants and tools.
Key Takeaway
Biocompatibility is the non-negotiable threshold for any medical component, serving as the primary barrier against adverse biological reactions.
| Standard | Description |
|---|---|
| ISO 10993 | Biological evaluation of medical devices |
| USP Class VI | Highest standard for plastics and polymers |
| RoHS/REACH | Chemical safety and restriction compliance |
| Bio-inertness | Total lack of interaction with human tissue |
Properly certified LSR ensures that your device remains safe for its entire intended service life within the human body.
How Does Medical Grade LSR Compare to Other Elastomers?

Medical Grade LSR consistently outperforms alternatives like TPE and HCR by offering a superior balance of thermal resistance, high-speed production, and low compression set. When comparing medical grade silicone for molding to other options, its ability to maintain mechanical properties across extreme temperature ranges is unmatched.
LSR vs. Thermoplastic Elastomers (TPE)
While TPEs are often cheaper and faster to process, they lack the chemical and thermal resistance of silicone. Make no mistake: TPE can melt or deform at temperatures where LSR remains completely stable.
- LSR maintains flexibility from -60°C to 200°C+.
- TPE has higher risk of leaching plasticizers.
- LSR offers far better long-term “spring back” (low compression set).
The HCR Comparison
High Consistency Rubber (HCR) is a traditional solid silicone, but it requires labor-intensive milling and manual loading. Liquid silicone rubber, however, uses a closed-loop automated system that virtually eliminates human-led contamination.
Key Takeaway
LSR is the high-performance “steroid” of the elastomer world, providing durability and purity that TPE and HCR simply cannot match in medical settings.
| Property | LSR | TPE | HCR |
|---|---|---|---|
| Heat Resistance | Excellent | Poor | Excellent |
| Bio-purity | Very High | Moderate | High |
| Processing | Automated | Fast | Manual |
| Compression Set | Very Low | High | Low |
The superior physical properties of LSR justify the higher material cost by extending product life and reducing failure rates.
What are the Different Classifications of Medical Silicone?
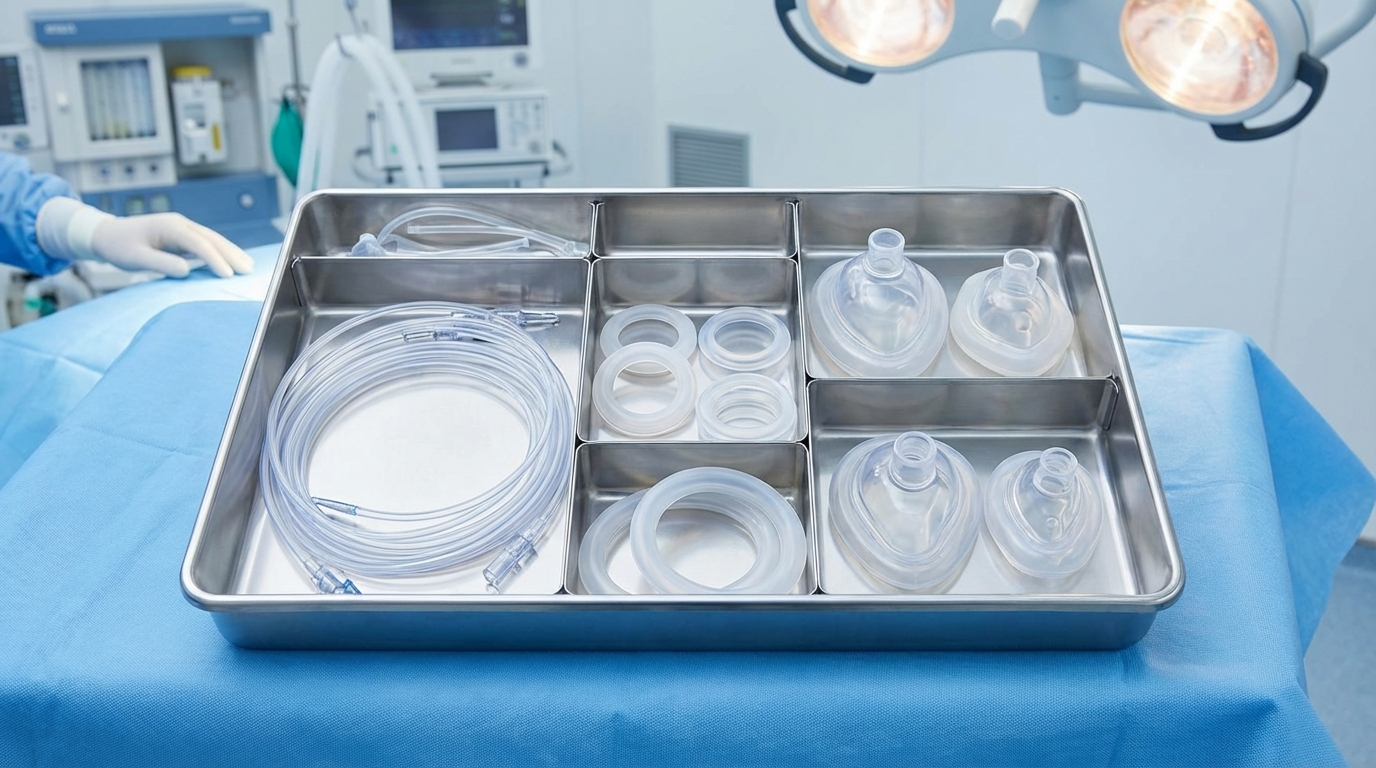
Medical silicone is classified into three primary categories—limited, prolonged, and permanent contact—based on how long the material interacts with the human body. Choosing the right medical grade silicone for molding depends entirely on your specific regulatory application.
Understanding Exposure Durations
Not all medical projects require the same level of material testing. For example, a skin-contacting mask doesn’t need the same chemical profile as a heart valve implant.
- Limited Contact: Skin or mucosal contact for 24 hours or less.
- Prolonged Contact: Exposure lasting between 24 hours and 30 days.
- Permanent Contact: Long-term implants exceeding 30 days of interaction.
Non-Implantable vs. Implantable Grades
Healthcare grade silicone is typically used for external devices and short-term internal exposure. However, long-term implantable LSR undergoes the most grueling physiochemical testing in the industry.
Key Takeaway
Accurately classifying your device’s contact duration is the first step in selecting a cost-effective yet compliant silicone grade.
| Grade | Contact Time | Common Application |
|---|---|---|
| Medical Non-Implant | < 24 Hours | Skin masks, handles |
| Healthcare Grade | 24 Hours – 30 Days | Catheters, IV components |
| Long-term Implant | > 30 Days | Pacemakers, stents |
Matching the material grade to the application prevents over-engineering costs while maintaining absolute regulatory compliance.
Why is Clean Room Molding Essential for Medical LSR?

Clean room molding is essential because it prevents microscopic airborne particulates and contaminants from being embedded in the medical grade silicone for molding during the curing process. In a clinical environment, even a single speck of dust can lead to inflammation or infection in a patient.
Controlling the Environment
Manufacturing takes place in ISO Class 7 or 8 environments where humidity, temperature, and air filtration are strictly monitored. But here is the kicker: the automation of the LSR process further reduces the risk of human skin oils or hair contaminating the batch.
- HEPA-filtered air circulation systems.
- Strict PPE requirements for all technicians.
- Integrated automated packaging within the clean zone.
Sterilization Readiness
Parts molded in a clean room are “sterilization-ready” for methods like Gamma radiation or ETO. This controlled start ensures that the final sterilization process is as effective as possible.
Key Takeaway
A clean room environment is the only way to guarantee the “clinical purity” required for parts used in surgery or fluid management.
| Clean Room Class | Max Particles (0.5µm) | Typical Application |
|---|---|---|
| ISO Class 7 | 352,000 / m³ | High-precision surgical tools |
| ISO Class 8 | 3,520,000 / m³ | Consumer medical devices |
| Standard Shop | Uncontrolled | Non-medical industrial parts |
Eliminating contamination at the source is much more effective than trying to “clean” a contaminated part post-production.
How Does the LSR Injection Molding Process Work?

The LSR injection molding process works by precisely mixing two liquid components and injecting them into a heated mold cavity where they rapidly cure into a solid state. Utilizing medical grade silicone for molding through this method allows for extreme repeatability and high-volume output.
The Closed-Loop Delivery System
The process begins with Part A and Part B being pumped from separate drums into a static mixer. You might be wondering how the ratio stays perfect; sophisticated metering systems ensure a strict 1:1 mix every single time.
- Automated dosing for zero material waste.
- Chilled injection barrels to prevent premature curing.
- High-pressure injection for complex, thin-walled geometries.
Rapid Curing Cycles
Unlike plastic, which must cool to harden, LSR requires heat to cross-link. The heated mold triggers a fast chemical reaction, allowing for cycles that are often measured in seconds rather than minutes.
Key Takeaway
The automated, closed-loop nature of LSR molding makes it the most consistent and hygienic manufacturing method for medical parts.
| Step | Action | Benefit |
|---|---|---|
| Mixing | 1:1 Dosing | Chemical consistency |
| Injection | High Pressure | Precision geometries |
| Curing | Heated Mold | Structural stability |
| De-molding | Automated | Flash-less, clean finish |
Automated LSR molding minimizes human error and provides the tightest tolerances required for medical seals and valves.
What are the Benefits of Overmolding in Medical Devices?

Overmolding offers the benefit of bonding soft, biocompatible silicone directly onto rigid plastic or metal substrates to create a single, unified component. By integrating medical grade silicone for molding with other materials, you can achieve ergonomic “soft-touch” surfaces that are permanently attached.
Ergonomics and Functionality
Multi-shot molding allows surgeons to have a better grip on instruments while maintaining a sterile surface. But there is a catch: the bond between the silicone and the substrate must be chemical-grade to prevent delamination during use.
- Ergonomic grips for surgical precision.
- Color-coding for tool identification.
- Shock absorption for delicate electronics.
Eliminating Contamination Gaps
Overmolding creates a seamless transition between materials, removing the tiny crevices where bacteria typically gather. This makes the device much easier to clean and sterilize between uses.
Key Takeaway
Overmolding combines the best of two worlds—rigidity and soft-touch safety—into one highly functional, sterile medical assembly.
| Benefit | Impact on Medical Device |
|---|---|
| Seamless Design | Zero bacteria traps |
| Chemical Bonding | No peeling or delamination |
| Soft-Touch | Improved clinician ergonomics |
| Integrated Seals | Waterproofing for electronics |
Strategic overmolding reduces assembly steps while significantly enhancing the safety and user experience of medical tools.
Which Medical Applications Benefit Most from LSR?

Applications that benefit most from LSR include those requiring high-precision seals, respiratory interfaces, and skin-contacting wearable monitors. The versatility of medical grade silicone for molding makes it the default choice for everything from anesthesia masks to insulin pump components.
Respiratory and Fluid Management
LSR’s clarity and flexibility make it perfect for valves and diaphragms that must react to tiny pressure changes. Here is the kicker: it doesn’t harden or crack over time, ensuring the device remains reliable for thousands of cycles.
- Resonator and anesthesia masks.
- One-way check valves and gaskets.
- Peristaltic pump tubing and connectors.
The Rise of Medical Wearables
As digital health expands, biocompatible straps and housings are in high demand. Silicone is hypoallergenic and sweat-resistant, making it comfortable for patients to wear 24/7.
Key Takeaway
The unique physical properties of LSR—flexibility, purity, and transparency—make it indispensable across nearly every specialized medical field.
| Category | Specific Application |
|---|---|
| Respiratory | Oxygen masks, nebulizer parts |
| Fluid Control | IV valves, syringe stoppers |
| Wearables | Heart rate monitor straps |
| Surgical | Instrument handles, trocar seals |
LSR’s broad application range allows medical OEMs to standardize on one material for multiple diverse product lines.
How Do Manufacturers Ensure Quality and Compliance?

Manufacturers ensure quality through a rigorous three-stage inspection system and full batch traceability for all medical grade silicone for molding. Every single part produced must be verifiable against its original material certification to ensure clinical safety.
The Three Pillars of Inspection
Quality isn’t just checked at the end; it is built into the process. But there is a catch: you must verify that your manufacturer holds an active ISO 13485 certification, which is specific to medical device quality systems.
- IQC (Incoming): Testing raw silicone batches before they ever touch the machine.
- IPQC (In-Process): Real-time monitoring of pressure, temperature, and dimensions.
- FQC (Final): Visual and functional testing of the finished part.
Precision Measurement Tools
Automated CCD visual inspection systems can catch surface defects that the human eye misses. This ensures that every seal is airtight and every dimension is within its specified tolerance.
Key Takeaway
A robust quality management system (QMS) is the only way to provide the audit trail necessary for regulatory approvals like the FDA or CE mark.
| Capability | Purpose |
|---|---|
| CCD Inspection | Automated defect detection |
| Tear Testing | Verification of material strength |
| Dimensional Check | Ensures perfect assembly fit |
| Lot Traceability | Links parts to raw material source |
Rigorous quality control converts a “part” into a “certified medical component” that you can trust in the operating room.
How Should You Select a Medical Silicone Partner?

You should select a partner based on their in-house tooling expertise, clean room capabilities, and deep understanding of medical grade silicone for molding regulations. A partner that offers DFM (Design for Manufacturing) support can save you months of trial-and-error during the prototyping phase.
Engineering and DFM Support
The best manufacturers don’t just “hit go” on your drawing; they analyze it for potential failure points. Here is the kicker: a slight adjustment in gate placement can be the difference between a high-yield run and a batch full of air bubbles.
- In-house mold making for faster iterations.
- Prototyping to mass production scalability.
- Assistance with material grade selection.
Compliance and Certifications
Always verify that your partner has the certifications to handle your market. If they lack ISO 13485 or FDA registration, they are likely not equipped to handle the documentation demands of a medical project.
Key Takeaway
Choosing a partner with specialized medical LSR experience reduces your time-to-market and mitigates your long-term regulatory risk.
| Criteria | Why it Matters |
|---|---|
| In-house Tooling | Tighter control over tolerances |
| ISO 13485 | Mandatory for medical quality |
| DFM Review | Prevents costly design errors |
| Clean Room | Prevents particulate contamination |
A true partner acts as an extension of your engineering team, ensuring that your medical vision is manufactured to the highest possible standards.
Conclusion
Navigating the complexities of medical manufacturing requires a material that balances uncompromising safety with high-performance physical properties. From the life-saving reliability of surgical valves to the patient-first comfort of smart wearables, Liquid Silicone Rubber has proven to be the premier solution for modern healthcare. By choosing a partner who understands the nuances of clean room production and rigorous quality compliance, you can launch your products faster and with total confidence.
Our vision is to empower medical device innovators with the precision engineering and certified materials they need to save lives and improve patient outcomes. If you are ready to elevate your project with industry-leading expertise, contact us today to start your technical consultation.
Frequently Asked Questions
Can I sterilize medical grade LSR multiple times?
Yes, you can. Medical grade LSR is highly resistant to standard sterilization methods including steam autoclaving, gamma radiation, and ETO without losing its physical properties or degrading.
What is the difference between Food Grade and Medical Grade silicone?
While both are high-purity, Medical Grade silicone undergoes more extensive biocompatibility testing according to ISO 10993. This ensures the material is safe for prolonged contact with human tissue and fluids.
Can medical grade silicone for molding be made in different colors?
Yes, it can. Manufacturers use medical-grade pigments to achieve specific Pantone colors, which is particularly useful for tool identification or brand aesthetics in consumer health products.
What is the standard hardness for medical LSR?
Medical LSR typically ranges from a soft 10 Shore A to a rigid 80 Shore A. This wide range allows engineers to select the exact level of flexibility required for their specific application.
Can medical grade LSR be used for permanent implants?
Yes, but you must specify “Long-term Implantable” grade. These specific materials undergo much more rigorous physiochemical testing than healthcare-grade silicones used for temporary devices.
