Silicone keypad models range from conductive carbon pill designs to non-conductive metal dome systems, each engineered for specific medical reliability. Many healthcare equipment designers face the challenge of interface failure due to fluid ingress or mechanical wear. This risk can lead to equipment downtime or even compromised patient safety during critical diagnostic procedures. Partnering with a premier silicone rubber keypad manufacturer provides you with the biocompatible, sealed, and durable solutions required to solve these engineering hurdles effectively.
Why choose silicone rubber keypads for medical gear?

You should choose silicone rubber keypads because they offer unmatched biocompatibility, chemical resistance, and the ability to withstand rigorous hospital sterilization cycles. As a specialized manufacturer, we recognize that medical interfaces must remain sterile and functional under heavy use. You need a material that does not harbor bacteria and remains stable when exposed to harsh disinfectants.
Is biocompatibility a factor in material selection?
Yes, it is the most critical factor for any user interface that comes into contact with medical staff or patients. You must ensure that the silicone used is medical-grade and hypoallergenic to meet safety regulations.
Here is why:
- Silicone is naturally resistant to microbial growth.
- It meets USP Class VI and ISO 10993 requirements.
- The material remains inert when exposed to bodily fluids.
- It withstands high-temperature autoclaving without degrading.
Key Takeaway
Silicone is the gold standard for medical interfaces due to its inherent resistance to contaminants and its ability to maintain integrity through extreme sterilization processes.
| Feature | Silicone Keypads | Traditional Plastics |
|---|---|---|
| Sterilization | Autoclave & Chemical Safe | Limited Resistance |
| Biocompatibility | High (USP Class VI) | Low to Moderate |
| Ingress Protection | IP67/IP68 Possible | Difficult to Seal |
| Durability | Millions of Cycles | Prone to Cracking |
Analysis: This comparison illustrates that silicone provides a superior safety profile and longer operational life in clinical environments compared to rigid plastics.
What are conductive silicone rubber keypads?
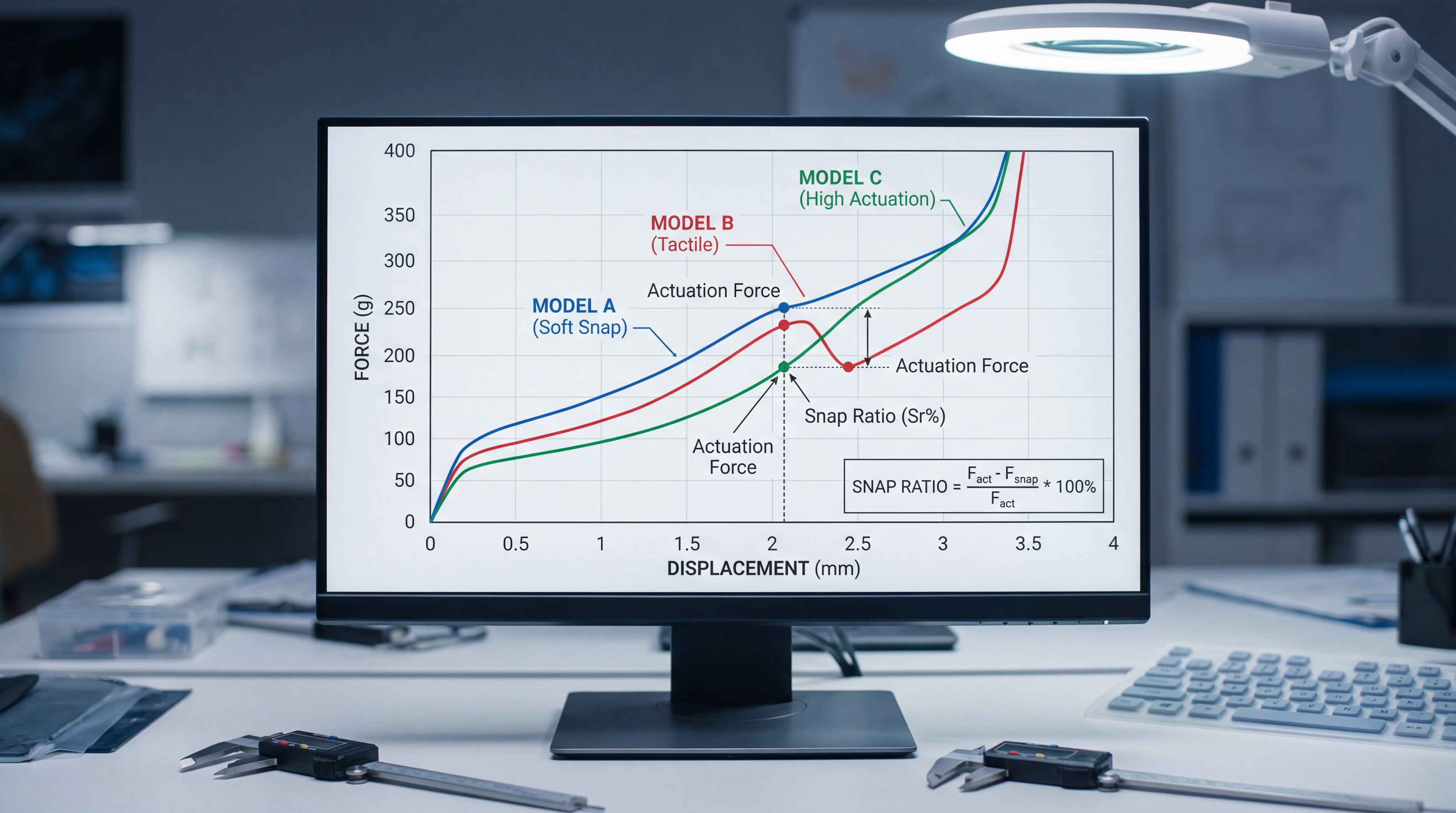
Conductive silicone rubber keypads use integrated carbon pills or gold-plated contacts to complete an electrical circuit when you press a key. A professional silicone rubber keypad manufacturer typically recommends these models for low-profile diagnostic equipment and handheld monitors. You can achieve a very slim device profile because the contact element is molded directly into the silicone mat.
How do carbon pills facilitate electrical contact?
Carbon pills are conductive disks permanently bonded to the bottom of the key actuators during the molding process. When you apply pressure, the pill bridges the gap between the traces on your printed circuit board (PCB).
Wait, there’s more:
- They offer low contact resistance for reliable signaling.
- Carbon pills are virtually wear-free over millions of actuations.
- They allow for silent operation in quiet hospital environments.
- You can choose different pill sizes to match your PCB layout.
Key Takeaway
Conductive models are ideal for space-constrained medical devices that require silent, reliable, and cost-effective electrical switching.
| Specification | Standard Value |
|---|---|
| Contact Resistance | < 100 Ohms |
| Life Expectancy | > 1,000,000 cycles |
| Pill Diameter | 2.0mm to 8.0mm |
| Travel Distance | 0.5mm to 3.0mm |
Analysis: The data confirms that conductive silicone components provide consistent electrical performance while maintaining a compact footprint suitable for portable medical tech.
How do non-conductive silicone keypads function?

Non-conductive silicone keypads function as mechanical actuators that physically press against an external switch, such as a metal snap dome, to provide tactile feedback. These are preferred when you need a clear, physical confirmation of a button press. These designs are highly effective for surgeons who need to operate equipment by feel while focusing on a patient.
Why is tactile response critical for surgeons?
Tactile response ensures that the user knows a command has been registered without needing to look at the device screen. This sensory “click” is vital for safety in high-pressure medical environments.
But that is not all:
- It prevents double-entry or missed commands.
- Metal domes provide a consistent “snap” feel.
- You can customize the actuation force to prevent accidental triggers.
- It enhances the overall premium feel of the medical instrument.
Key Takeaway
Non-conductive models paired with metal domes offer the high-tactile feedback necessary for precision-critical medical applications.
| Metric | Non-Conductive (Metal Dome) |
|---|---|
| Tactile Feel | Sharp, crisp “click” |
| Actuation Force | 250g – 500g |
| Switch Travel | 0.3mm – 0.5mm |
| Auditory Feedback | Audible “snap” |
Analysis: The sharp tactile and auditory feedback provided by these models reduces user error by confirming inputs through multiple sensory channels.
How do mechanical properties differ between types?

Mechanical properties differ primarily in the actuation force, the travel distance of the key, and the resulting snap ratio that you feel. Your chosen silicone rubber keypad manufacturer must rigorously test these parameters to ensure the interface meets your ergonomic requirements. You have the flexibility to adjust the hardness of the silicone to change how the keys respond to touch.
Is keytop travel distance a major differentiator?
Yes, because travel distance determines the “softness” or “snappiness” of the user interface. Conductive models usually have longer travel, while metal dome models provide a shorter, more immediate stroke.
Consider this:
- Longer travel (1.5mm+) offers a traditional “button” feel.
- Short travel (<0.5mm) is better for rapid, repetitive data entry.
- Precise webbing design prevents keys from wobbling or sticking.
- Consistent travel ensures a uniform experience across the entire keypad.
Key Takeaway
Understanding mechanical differences allows you to choose between a soft, quiet interface and a sharp, tactile one depending on the clinical setting.
| Feature | Conductive Models | Non-Conductive Models |
|---|---|---|
| Operating Force | 60g – 200g | 240g – 2,000g |
| Snap Ratio | 40% – 60% | 50% – 80% |
| Mechanical Life | High | Very High |
| Return Force | 30g – 80g | 50g – 150g |
Analysis: By balancing force and travel distance, engineers can customize the user experience to match specific task requirements within the hospital.
What sealing advantages do medical keypads offer?

Medical silicone keypads offer superior sealing through a monolithic mat design that prevents liquids, dust, and blood from reaching the internal electronics. A dedicated producer can design a “wrap-around” lip that acts as a permanent gasket. This ensures that you can wash or disinfect the device without risking a short circuit.
Can these keypads prevent fluid ingress?
Yes, because the silicone mat is molded as a single piece, there are no seams for fluids to penetrate. You can achieve IP67 or even IP68 ratings for devices used in emergency rooms or labs.
Here is the kicker:
- Elimination of crevices prevents bacterial colonization.
- The material is naturally hydrophobic, repelling water and chemicals.
- It protects sensitive PCBs from high-pressure cleaning.
- Silicone maintains its seal even under thermal expansion.
Key Takeaway
Integrated sealing is a primary advantage of silicone keypads, protecting both the electronics and the hygienic integrity of the medical device.
| Hazard Type | Protection Level | Benefit |
|---|---|---|
| Isopropyl Alcohol | Excellent | Frequent disinfection safe |
| Bleach Solutions | High | Deep cleaning compatibility |
| Dust/Particulates | Complete Seal | Cleanroom compliant |
| Saline/Blood | Excellent | Easy to wipe and sanitize |
Analysis: The monolithic structure effectively shields internal components from common biohazards and corrosive cleaning agents.
How does operation force impact user experience?

Operation force dictates how much pressure you must apply to register a keypress, which directly affects user fatigue and input accuracy. Collaborating with an expert team allows you to fine-tune these forces for specific buttons on your device. For instance, you might want a higher force for a “Power” button to prevent accidental shutoffs.
How to avoid wobble during off-center presses?
You can avoid key wobble by designing wide actuation bases and optimizing the thickness of the silicone webbing. This ensures that even if you press the edge of a key, it travels straight down to make contact.
Think about this:
- Uniform force distribution improves user confidence.
- Optimized webbing reduces the “mushy” feel of low-quality keypads.
- Specific gram-force tuning caters to different user demographics.
- High-quality molds ensure every key feels identical.
Key Takeaway
Precision tuning of operation force and key stability is essential for creating a professional and reliable medical interface.
| Button Function | Recommended Force | Purpose |
|---|---|---|
| Data Entry/Arrows | 80g – 120g | Ease of use/Speed |
| Start/Action | 150g – 250g | Intentional press |
| Emergency/Power | 300g+ | Prevent accidents |
| Soft Keys | 60g – 100g | Light touch response |
Analysis: Strategic force allocation ensures critical functions require deliberate action while routine inputs remain effortless.
Why is ISO 13485 vital for medical production?
ISO 13485 is vital because it ensures that the manufacturing process follows a strict quality management system tailored for the medical industry. This certification proves that the factory can manage risks and maintain consistent production standards. You need this level of compliance to ensure your device meets global regulatory requirements.
Does cleanroom molding reduce contamination?
Yes, manufacturing in a Class 100,000 cleanroom prevents airborne particulates from being trapped in the silicone during the curing process. This is essential for maintaining the aesthetic and functional purity of medical components.
The reason is simple:
- It minimizes the risk of bioburden on the keypad surface.
- Cleanroom environments ensure higher cosmetic yields for clear silicone.
- It meets the stringent requirements of FDA-regulated devices.
- Traceability is maintained from raw material to finished part.
Key Takeaway
Working with an ISO 13485 certified manufacturer guarantees that your keypads are produced under the highest safety and quality protocols.
| Standard | Focus Area | Requirement |
|---|---|---|
| ISO 13485 | Medical QMS | Risk management & Traceability |
| FDA Grade | Material Safety | Non-toxic, food/medical safe |
| RoHS/REACH | Chemical Safety | No hazardous substances |
| ISO 9001 | General Quality | Process consistency |
Analysis: Compliance with these standards provides the necessary documentation and safety assurance required for medical device market entry.
Can custom finishes enhance keypad durability?

Custom finishes like polyurethane (PU) coatings and epoxy resins significantly enhance the resistance of keypads to mechanical abrasion and chemical cleaning. A versatile silicone rubber keypad manufacturer offers these treatments to protect the printed graphics on your device. You can ensure that the “Start” and “Stop” labels remain visible even after years of daily hospital use.
Are laser-etched graphics better for long use?
Yes, laser etching involves painting a translucent silicone mat and then using a laser to remove the paint in the shape of your icons. This creates symbols that are part of the coating and cannot be rubbed off.
But there is more:
- Laser etching allows for crisp, backlit legends.
- PU coatings provide a “silk-feel” that resists oils and sweat.
- Epoxy resins can create a hard, plastic-like feel on keytops.
- Antimicrobial additives can be integrated into the top coating.
Key Takeaway
Custom surface treatments are necessary to protect the longevity of your device’s branding and operational labels in high-wear environments.
| Finishing Type | Abrasion Resistance | Chemical Resistance |
|---|---|---|
| Standard Ink | Moderate | Low |
| PU Coating | High | Moderate |
| Epoxy Resin | Very High | High |
| Laser Etching | Permanent | High |
Analysis: Specialized coatings ensure that vital instructional legends remain legible throughout the entire lifespan of the medical equipment.
What quality standards should a manufacturer meet?

A manufacturer should meet FDA, LFGB, and REACH standards while performing comprehensive life-cycle and actuation force testing. Ensuring that the factory follows these protocols protects your company from liability and potential product recalls. You must verify that the raw materials are sourced from reputable suppliers to ensure consistency.
How is cycle life testing performed?
Engineers use automated pneumatic or electronic probes to depress the keys millions of times while monitoring for material fatigue. They also check for electrical continuity to ensure the conductive pills remain functional.
Keep this in mind:
- Life testing often exceeds 5 million actuations for medical gear.
- Environmental chambers test performance in high humidity and heat.
- Salt spray tests ensure the keypad survives coastal or saline environments.
- Snap ratio testing confirms the tactile feel doesn’t degrade over time.
Key Takeaway
Rigorous testing protocols ensure that your silicone interface will outlast the electronic components of the medical device itself.
| Test Type | Objective | Pass Criteria |
|---|---|---|
| Force Displacement | Measure tactile feel | Within ±15% of spec |
| Resistance Test | Check electrical path | < 100 Ohms (Conductive) |
| Life Cycle Test | Determine durability | No cracks at 1M+ cycles |
| Dimensional Check | Verify fitment | ±0.1mm tolerance |
Analysis: Detailed validation steps eliminate manufacturing variability, ensuring every unit shipped meets stringent medical performance requirements.
How to select the right silicone keypad partner?

You should select a partner based on their engineering depth, in-house tooling capabilities, and their specific experience with the rigorous requirements of the medical sector. Choosing the right partner ensures that your project moves smoothly from the initial CAD design to mass production. You need a team that can provide micro-molding for the small, intricate devices common in modern healthcare.
Should they offer in-house tooling services?
Yes, in-house tooling allows for faster design iterations and ensures that the mold precision is maintained throughout the product lifecycle. This reduces your time-to-market and lowers overall development costs.
Here is why it matters:
- Rapid prototyping allows for immediate ergonomic testing.
- Tooling modifications can be made on-site within hours.
- You have a single point of accountability for quality.
- Mold maintenance is handled automatically by the manufacturer.
Key Takeaway
A vertically integrated partner offers the technical agility and quality control necessary for rapid, compliant medical device development.
| Benefit | In-House Tooling | Outsourced Tooling |
|---|---|---|
| Lead Time | 7-14 Days | 21-30 Days |
| Modifications | Instant | Multi-day shipping |
| Precision Control | Direct | Indirect |
| Cost Efficiency | High | Low |
Analysis: Owning the tooling process directly translates to higher manufacturing accuracy and a significantly reduced time-to-market for complex medical interfaces.
Our vision is to empower medical innovation by providing the most reliable, precise, and hygienic user interfaces in the world. By addressing the problems of durability and sterilization, we ensure that healthcare professionals can focus on patient care without worrying about equipment failure. To start your next project with a trusted expert, please contact us to discuss your specific medical-grade requirements.
FAQ
Can I customize the color and shape of the keys?
Absolutely, silicone allows for complete freedom in geometry and can be color-matched to any Pantone reference you provide. This flexibility is perfect for color-coding critical medical functions.
What’s the best coating for frequent chemical cleaning?
A polyurethane (PU) coating is typically the best choice because it provides a strong barrier against alcohols and disinfectants. It also maintains a smooth, non-stick surface that is easy to wipe down.
How do I know if my design needs conductive pills?
Consult your PCB specifications; if your board has open contact traces intended for a short, you need conductive pills. If you are using individual tactile switches on the board, a non-conductive actuator is the right choice.
Can I get rapid prototypes before mass production?
Yes, most top manufacturers offer liquid silicone rubber (LSR) or compression molded prototypes within 1-2 weeks. This allows you to verify the tactile feel and fitment before investing in multi-cavity production tools.
What’s the best way to ensure liquid resistance?
The best method is to design the keypad with an integrated perimeter seal or “apron” that overlaps the device housing. This creates a physical barrier that prevents any fluid from reaching the electronics.

