Medical-grade silicone is a specialized synthetic elastomer specifically engineered to meet stringent biocompatibility and safety standards for healthcare applications. Selecting an inferior material for medical devices can lead to product failure, regulatory rejection, and significant patient risk. By utilizing high-purity medical grade silicone, you ensure your components are non-reactive, durable, and fully compliant with international clinical requirements.
1. What is medical-grade silicone?
Medical-grade silicone is a high-purity synthetic polymer that has been extensively tested for safe use in human contact and clinical environments. This material is distinguished from industrial or food grades by its extreme chemical stability and low levels of extractables.
Is chemical stability important?
Chemical stability is the foundation of patient safety in healthcare settings. Because this material is inert, it does not leach harmful chemicals or react with biological fluids during use.
But that’s not all. You must also consider that these polymers maintain their physical integrity even when exposed to harsh medications or aggressive cleaning agents.
What are the key regulatory standards?
Regulatory compliance ensures that the material meets the highest benchmarks for toxicity and biocompatibility. The most common standard is USP Class VI, which involves rigorous systemic and intracutaneous testing.
- USP Class VI certification for biocompatibility.
- ISO 10993 compliance for medical device safety.
- Platinum-cured systems for minimal byproducts.
- FDA 21 CFR 177.2600 for food-contact safety.
| Standard | Scope | Primary Requirement |
|---|---|---|
| USP Class VI | Systemic Toxicity | No adverse biological response in vivo |
| ISO 10993 | Biocompatibility | Cytotoxicity and sensitization testing |
| Platinum Cure | Purity | No peroxide byproducts or blooming |
This data confirms that choosing a material with these specific certifications is non-negotiable for medical device legal compliance.
Key Takeaway: Medical-grade silicone provides a foundation of chemical stability and regulatory compliance essential for avoiding systemic toxicity in patients.
2. Why is biocompatibility vital for safety?
Biocompatibility is vital because it ensures that medical grade silicone does not trigger an immune response, irritation, or toxic reaction when it touches human tissue. This characteristic is what makes the material the gold standard for long-term implants and skin-contact wearables.
How does the immune system react?
The human body is highly sensitive to foreign materials and often attempts to reject them. Because medical silicone is fundamentally inert, the body’s immune cells do not recognize it as a threat.
Think about it. Without this “biocompatible” status, a simple heart valve or catheter could cause life-threatening inflammation or rejection symptoms.
Does it resist microbial growth?
Healthcare environments are prone to bacterial contamination, making material resistance a top priority. The molecular structure of high-purity silicone naturally resists the formation of biofilms and fungal growth.
- Inherent resistance to bacteria and fungi.
- Hydrophobic surface to repel fluids.
- Non-porous structure to prevent absorption.
- Sterile-ready surface for surgical use.
| Property | Benefit | Patient Outcome |
|---|---|---|
| Inertness | Low immune response | Reduced inflammation |
| Bio-resistance | No microbial growth | Lower infection risk |
| Hypoallergenic | No latex or BPA | Safe for sensitive skin |
The inert properties shown here highlight why silicone remains the safest choice for prolonged contact with biological systems.
Key Takeaway: Biocompatibility minimizes patient risk by preventing immune rejection and inhibiting the growth of hospital-acquired infections.
3. What are the key advantages of this material?

The key advantages of medical grade silicone include its exceptional thermal stability, chemical resistance, and the ability to maintain its mechanical properties over millions of cycles. These features allow components to withstand the most demanding clinical conditions without degrading.
How does it handle extreme temperatures?
Medical components often face temperature extremes during both operation and sterilization cycles. This elastomer remains flexible at -50°C and stable up to 250°C, outperforming most organic rubbers.
The best part? It maintains this flexibility without becoming brittle or melting, ensuring long-term device reliability.
Is mechanical durability a factor?
Durability is critical for seals, gaskets, and valves that must operate repeatedly. The material has a low compression set, meaning it returns to its original shape after being compressed for long periods.
- High tensile strength for surgical tools.
- Excellent elongation for flexible tubing.
- Vibration damping for diagnostic machinery.
- Long shelf life of 20+ years.
| Metric | Performance Range | Application |
|---|---|---|
| Temperature Range | -50°C to +250°C | Autoclave sterilization |
| Hardness (Shore A) | 10 to 80 | From soft cushions to rigid seals |
| Elongation | Up to 800% | Stretchable tubing and membranes |
The wide range of physical properties makes silicone incredibly versatile for both structural and flexible medical components.
Key Takeaway: High thermal and mechanical durability ensure that medical components perform reliably through repeated usage and sterilization cycles.
4. Where is medical-grade silicone used?
Medical grade silicone is used across a vast spectrum of healthcare products, including wearable and medical accessories, diagnostic equipment, and life-saving surgical instruments. Its versatility allows it to be manufactured as thin-walled tubing, complex valves, or soft skin interfaces.
What are common surgical applications?
In the operating room, precision and sterility are the only things that matter. Silicone is the primary material for catheters, drainage tubes, and surgical instrument handles due to its grip and sterilizability.
Here is the kicker. Its transparency allows surgeons to monitor fluid flow in real-time during critical procedures.
How is it used in diagnostics?
Modern diagnostic machines like MRI and ventilators rely on silicone for vibration damping and airtight sealing. Without high-purity seals, these machines could leak oxygen or fail to provide accurate readings.
- Ventilator masks and diaphragms.
- Catheters and IV delivery systems.
- Damping gaskets for imaging equipment.
- Feeding tubes and wound drains.
| Application Type | Common Components | Primary Requirement |
|---|---|---|
| Fluid Management | Tubing, Catheters | Transparency & Purity |
| Diagnostic Gear | Gaskets, Keypads | Durability & Cleanliness |
| Patient Care | Masks, Feeding Tubes | Comfort & Biocompatibility |
The breadth of applications demonstrates that silicone is an indispensable material in almost every department of a modern hospital.
Key Takeaway: From fluid delivery to high-tech diagnostics, silicone provides the necessary purity and performance for a wide range of medical devices.
5. How does cleanroom manufacturing work?

Cleanroom manufacturing involves producing medical grade silicone components in a controlled environment to minimize airborne particles and biological contaminants. This process is essential for ensuring that every part leaves the factory with a low bio-burden.
What are ISO environment standards?
ISO Class 8 (also known as Class 100,000) is the standard environment for most medical silicone molding. This classification limits the number of particulates per cubic meter of air to ensure part purity.
Why does this matter? Even a single microscopic dust particle trapped in a heart valve can lead to catastrophic failure once implanted.
How is contamination prevented?
Strict protocols are followed for both machinery and personnel to maintain a sterile workflow. Technicians wear specialized attire, and raw materials are handled in filtered air stages to prevent cross-contamination.
- HEPA-filtered air systems for particulate control.
- Technicians in full protective hazmat-style suits.
- Stainless steel molds to prevent rust and leaching.
- Automated injection systems to reduce human contact.
| ISO Class | Particle Limit (0.5µm) | Medical Usage |
|---|---|---|
| ISO Class 7 | 352,000 / m³ | Highly sensitive implants |
| ISO Class 8 | 3,520,000 / m³ | Standard medical molding |
| General Factory | Uncontrolled | Non-medical industrial parts |
This comparison clarifies why specialized cleanroom facilities are mandatory for any reputable medical device component manufacturer.
Key Takeaway: Cleanroom production in ISO-standard environments is the only way to guarantee the low bio-burden required for clinical safety.
6. Which molding processes are most effective?
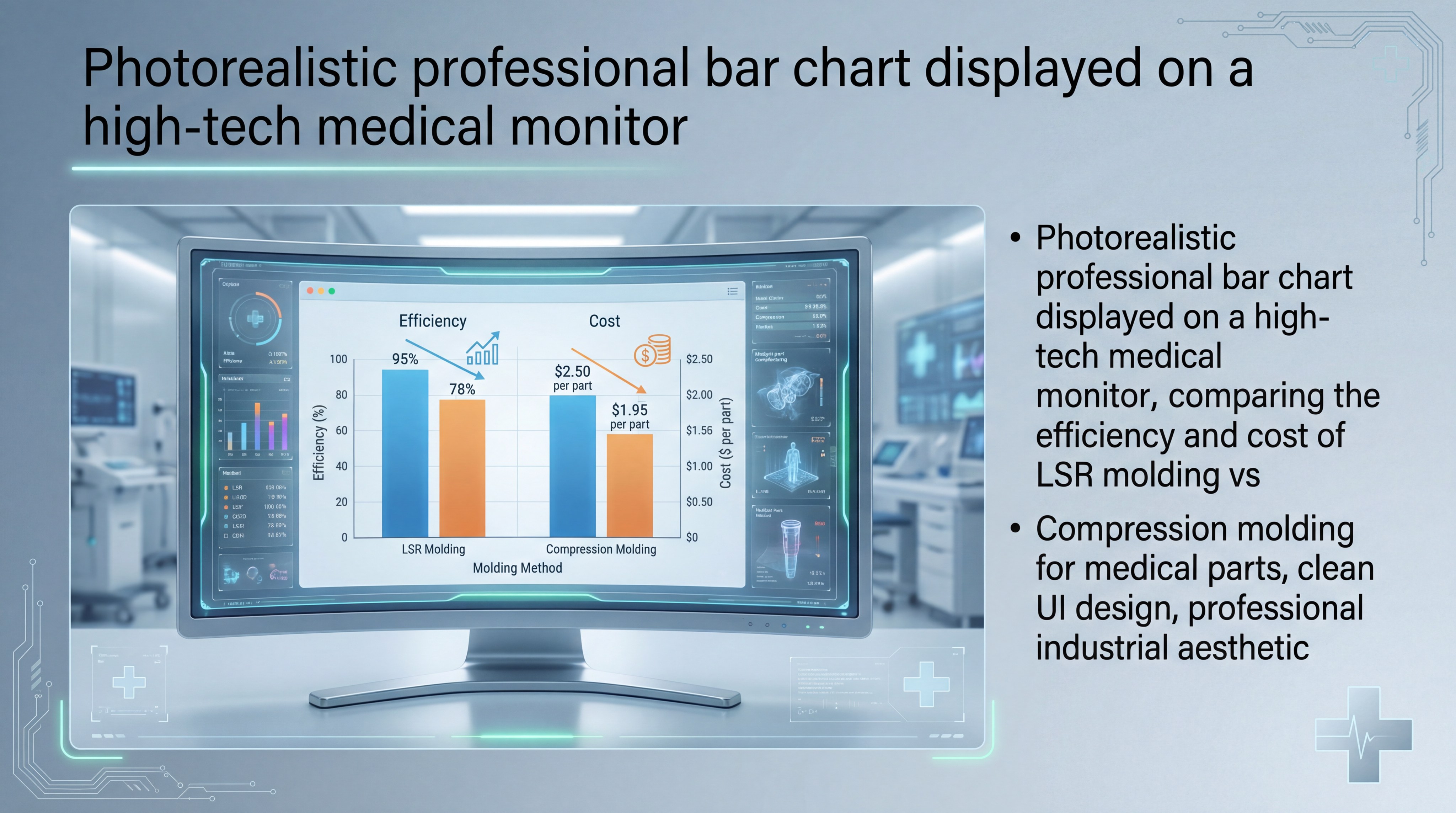
Liquid Silicone Rubber (LSR) injection molding is the most effective process for high-precision, high-volume medical grade silicone parts. This automated process ensures consistency and allows for complex geometries that other methods cannot achieve.
Why choose LSR injection molding?
LSR molding uses a liquid two-part system that is pumped into a heated mold for rapid curing. This method is ideal for parts requiring tight tolerances and intricate details, such as thin membranes or micro-valves.
The results speak for themselves. Automated LSR molding reduces human error and produces flash-free parts that require no secondary finishing.
When is compression molding used?
For simpler shapes or lower-volume production, compression molding remains a cost-effective alternative. It is often used for larger gaskets or thicker mats where the high speed of LSR is not required.
- LSR Injection for high-precision and high volume.
- Compression molding for simpler, thicker parts.
- Overmolding to bond silicone to plastic or metal.
- Micro-molding for extremely small medical components.
| Method | Precision Level | Best For |
|---|---|---|
| LSR Injection | Ultra-High (±0.01mm) | Complex valves and tubing |
| Compression | Moderate | Gaskets and handles |
| Overmolding | High | Surgical tools and wearables |
As the efficiency chart illustrates, LSR molding provides the best balance of speed and precision for modern healthcare manufacturing.
Key Takeaway: Selecting the right molding process—primarily LSR for complexity—ensures both cost-efficiency and high-tolerance performance.
7. Is medical-grade silicone safe for skin?

Yes, medical grade silicone is exceptionally safe for skin contact because it is hypoallergenic, latex-free, and contains no sensitizing chemicals. These properties make it the ideal choice for long-term wearable health monitors and respiratory masks.
Is it better than latex?
Unlike natural rubber latex, which can cause severe allergic reactions in many patients, silicone is universally tolerated. It does not cause the rashes or dermatitis often associated with lower-grade elastomers.
You might be wondering. Does it stay comfortable over time? Because it is breathable and soft, it prevents skin maceration even when worn 24/7.
How is skin safety tested?
Safety for skin contact is validated through specific ISO 10993 tests for irritation and sensitization. These tests ensure the material does not cause a reaction when exposed to sweat or body oils.
- Latex-free and BPA-free composition.
- Validated via ISO 10993-10 skin irritation tests.
- Soft-touch texture for patient comfort.
- Resistant to degradation from skin oils and sweat.
| Test Metric | Result for Silicone | Patient Benefit |
|---|---|---|
| Sensitization | Non-sensitizing | No allergic reactions |
| Irritation | Non-irritating | Safe for sensitive skin |
| Toxicity | Non-toxic | No leaching into bloodstream |
The data confirms that silicone is the premier material for any device that requires prolonged or repeated contact with human skin.
Key Takeaway: Hypoallergenic properties and rigorous irritation testing make silicone the safest material for skin-contact medical wearables.
8. How do you sterilize medical silicone?

You can sterilize medical grade silicone using multiple methods, including steam autoclaving, ethylene oxide (EtO) gas, and gamma radiation. Its ability to withstand these processes without physical degradation makes it highly reusable and sustainable.
Is steam autoclaving safe?
Steam autoclaving is the most common sterilization method in hospitals due to its speed and effectiveness. Since silicone handles temperatures up to 250°C, it can undergo hundreds of autoclave cycles without losing its shape.
Here is the secret. While other plastics might warp or crack, silicone remains flexible and functional.
What about radiation methods?
Gamma and E-beam radiation are often used for single-use, pre-packaged devices like catheters. Silicone is compatible with these methods and does not become brittle or yellow after exposure.
- Autoclave (Steam) for reusable instruments.
- Ethylene Oxide (EtO) for delicate electronics.
- Gamma Radiation for pre-packaged disposables.
- Dry heat sterilization for high-heat resilience.
| Method | Process Type | Silicone Compatibility |
|---|---|---|
| Autoclave | Heat & Pressure | Excellent (Reusable) |
| EtO Gas | Chemical | Excellent (No residue) |
| Gamma | Radiation | Excellent (No embrittlement) |
The versatility shown here proves that silicone is compatible with every major sterilization modality currently used in modern healthcare.
Key Takeaway: Exceptional resilience across various sterilization methods makes silicone a cost-effective and sustainable choice for reusable medical tools.
9. What should you look for in a supplier?

When choosing a supplier for medical grade silicone, you should prioritize ISO 13485 certification, in-house tooling capabilities, and a proven track record in medical manufacturing. A qualified supplier acts as a technical partner rather than just a parts manufacturer.
Why is ISO 13485 critical?
ISO 13485 is a quality management system specifically for the medical device industry. It ensures that the supplier has full traceability, strict quality control, and risk management protocols in place.
Look at it this way. If a supplier lacks this certification, you have no guarantee that their processes meet medical safety laws.
Does in-house tooling matter?
In-house mold making allows for faster prototyping and ensures that the molds are optimized for medical tolerances. It also ensures better protection for your intellectual property and design specifications.
- Full material traceability (lot and batch).
- In-house DFM (Design for Manufacturing) reviews.
- ISO Class 8 cleanroom molding facilities.
- Support for regulatory documentation and audits.
| Requirement | Why it Matters | Risk if Missing |
|---|---|---|
| ISO 13485 | Legal Compliance | Regulatory rejection |
| In-House Tooling | Precision & Speed | Part failure / Delay |
| Cleanroom | Purity | Bio-burden contamination |
Choosing a supplier based on these criteria ensures your medical project is handled with the precision and documentation required for success.
Key Takeaway: Certifications like ISO 13485 and in-house engineering expertise are non-negotiable requirements for a medical silicone supplier.
10. How does it compare to food-grade options?

While both are high-quality, medical grade silicone must meet far stricter biocompatibility standards (like USP VI) compared to food-grade options (FDA 21 CFR). Food-grade focuses on taste and smell transfer, whereas medical-grade focuses on biological safety inside the body.
Can I swap one for the other?
Substituting food-grade for medical-grade in a clinical device is a major safety and legal risk. Food-grade materials may contain trace impurities that are safe to digest but harmful when in contact with open wounds or blood.
Don’t take the risk. Medical-grade systems are specifically formulated to be the “cleanest” versions of the polymer available.
What is the curing difference?
Most medical silicones are platinum-cured, which results in a material with no peroxide byproducts and higher clarity. Food-grade options sometimes use peroxide curing, which can leave behind a white “bloom” on the surface.
- Medical: USP Class VI / ISO 10993.
- Food: FDA 21 CFR 177.2600.
- Medical: Higher purity, platinum-cured only.
- Food: Peroxide or platinum curing allowed.
| Feature | Food-Grade | Medical-Grade |
|---|---|---|
| Primary Goal | Non-toxic for digestion | Non-toxic for the body |
| Curing Method | Peroxide or Platinum | Strictly Platinum (usually) |
| Testing | Extraction tests | Biocompatibility tests |
This comparison highlights that only medical-grade material provides the level of safety validation required for modern healthcare devices.
Key Takeaway: Medical-grade silicone offers superior purity and biological validation that cannot be matched by standard food-grade elastomers.
Conclusion
Medical-grade silicone remains the premier material for the healthcare industry due to its unparalleled biocompatibility, durability, and versatility. By understanding the rigorous standards of cleanroom manufacturing and regulatory compliance, you can make informed decisions that protect both your business and your patients. Our vision is to provide high-precision silicone solutions that drive the future of medical technology. To start your next project with a trusted manufacturing partner, please contact us today.
FAQ
Can I use food-grade silicone for medical devices?
No. Food-grade lacks the biocompatibility certifications (USP Class VI) required for clinical safety and patient contact.
Is all medical silicone platinum-cured?
Generally, yes. Platinum curing is preferred because it leaves no peroxide byproducts, ensuring the material remains pure and non-reactive.
How do I know if my supplier is truly medical-grade?
Ask for their ISO 13485 certification and USP Class VI material test reports to verify their quality claims.
What is the best sterilization method for reusable silicone?
Steam autoclaving is the industry standard for reusables because silicone can withstand high heat and pressure repeatedly.
Does medical silicone contain latex or BPA?
No. High-purity medical silicone is inherently free of latex, BPA, and phthalates, making it hypoallergenic and safe for all patients.
