Cleaning white shoes effectively requires a comprehensive understanding of the high-performance polymers, such as silicone-based finishes and protective coatings, that maintain material integrity against environmental stressors. You often find that premium footwear loses its luster due to deep-seated industrial pollutants and the yellowing effects of oxidation on synthetic surfaces. This frustration is agitated by the reality that standard cleaning agents often fail to remove stubborn stains, while the shedding of microplastics from silicone in low-grade accessories creates an invisible environmental burden. We provide the technical expertise needed to navigate these material complexities, ensuring your products retain their functional and aesthetic excellence through superior manufacturing insights.
What is the true chemical nature of silicone?

Silicone is a sophisticated synthetic polymer defined by a unique backbone of alternating silicon and oxygen atoms, known as a siloxane chain. This inorganic core distinguishes it from carbon-based plastics and prevents the typical fragmentation associated with microplastics from silicone found in standard hydrocarbons. You can observe its purity in its resistance to UV radiation and moisture, making it an ideal candidate for high-end industrial applications. The molecular stability provided by this Si-O-Si structure ensures that the material remains flexible without the need for volatile plasticizers.
The role of siloxane in molecular structure
The siloxane backbone is the foundational element that gives silicone its legendary flexibility and thermal stability across diverse environments. Unlike carbon-carbon bonds, these silicon-oxygen bonds are stronger and more resistant to chemical attack from common solvents or industrial cleaners. This chemical resilience is why you choose silicone for components that must survive where traditional organic polymers would simply fail.
How silicon dioxide transforms into synthetic polymers
The manufacturing journey begins with silicon dioxide, a naturally occurring mineral typically sourced from high-quality silica sand. Through a multi-stage chemical synthesis, this sand is converted into various liquid, gel, or solid forms of silicone through the addition of organic groups like methyl or vinyl. This hybrid nature allows the material to bridge the gap between inorganic minerals and organic plastics.
Here is the bottom line:
- High thermal stability up to 250°C.
- Strong resistance to oxidative degradation.
- Low surface energy for easy cleaning.
- Inherent water repellency (hydrophobicity).
Is the chemical structure stable?
The inherent stability of the siloxane bond means that the material does not easily break down into hazardous subunits during regular use. While traditional polymers might degrade into harmful particulates, high-quality silicone maintains its physical properties for decades under normal operating conditions. You can rely on this stability to prevent the premature failure of seals, gaskets, and protective shoe coatings.
Key Takeaway: The unique inorganic backbone of silicone provides a level of durability and chemical resistance that far exceeds traditional petroleum-based materials.
| Property | Silicone (Siloxane) | Traditional Plastic (Hydrocarbon) |
|---|---|---|
| Backbone | Silicon-Oxygen | Carbon-Carbon |
| UV Resistance | Excellent | Poor to Moderate |
| Thermal Range | -60°C to 250°C | 0°C to 100°C |
| Chemical Inertness | High | Variable |
This comparison highlights why the siloxane structure is preferred for extreme durability requirements.
Why is silicone often confused with traditional plastic?

Silicone is frequently confused with plastic because it shares physical traits like flexibility and moldability, yet it is fundamentally a silica-based inorganic polymer. When you analyze the environmental impact, it is crucial to understand that microplastics from silicone are structurally different from the toxic particles shed by petroleum-derived plastics. This distinction is vital for industries moving toward sustainable manufacturing and reduced environmental footprints. Despite their similar appearance on a laboratory workbench, their chemical reactivity and lifecycle characteristics are worlds apart.
Comparing petroleum-based and silica-based materials
Petroleum-based plastics rely on finite fossil fuels and are characterized by long chains of carbon atoms that can be highly reactive. Silica-based silicone, conversely, utilizes one of the most abundant minerals on Earth, leading to a material that is significantly more inert. You will find that silicone does not possess the same leaching potential as plastics that contain BPA or phthalates.
Key differences in chemical reactivity
The chemical reactivity of silicone is exceptionally low, which prevents it from bonding with or reacting to most substances it encounters. Plastics often react with heat, releasing fumes or changing their internal structure, whereas silicone maintains its state. But it gets better: this inertness also means that silicone surfaces are naturally antimicrobial and easier to sterilize than many plastic alternatives.
- Carbon-based polymers are prone to hardening.
- Silicone maintains elasticity indefinitely.
- Plastic requires additives for flexibility.
- Silicone is inherently flexible at a molecular level.
Why do they look so similar?
Modern manufacturing techniques can make silicone look and feel exactly like various types of rubber or plastic. This visual similarity is why you must rely on chemical testing or manufacturer certifications to verify the material’s true nature. Using the wrong material can lead to catastrophic failure in applications involving high heat or aggressive chemicals.
Key Takeaway: While they may share aesthetic qualities, the inorganic origin of silicone makes it a safer and more durable choice than conventional plastics.
| Feature | Silicone | Traditional Plastic |
|---|---|---|
| Primary Resource | Silica Sand | Crude Oil |
| Flexibility Source | Molecular Structure | Added Plasticizers |
| Environmental Shedding | Siloxane Fragments | Microplastics |
| Toxicity Profile | Low/Inert | Potential Leaching |
Understanding these distinctions is the first step toward optimizing your material selection process.
How does silicone compare to silicone rubber?

Silicone rubber is a specific, cross-linked form of silicone that has been engineered to exhibit high elasticity and mechanical strength. While all silicone rubber is made from silicone, not all silicone is rubber; it can also exist as oils, resins, and gels that do not shed microplastics from silicone in the same way solid elastomers might. You will notice that silicone rubber has a “memory,” allowing it to return to its original shape after being stretched or compressed. This property is achieved through a process called vulcanization or curing, which creates a three-dimensional network of chemical bonds.
Understanding the cross-linking process
Cross-linking involves the use of a catalyst—typically platinum or peroxide—to link individual polymer chains together. This process transforms the material from a viscous liquid or soft paste into a durable, resilient solid. You can adjust the density of these cross-links to control the final hardness and stretchability of the material for specific industrial needs.
Liquid silicone versus flexible rubber forms
Liquid Silicone Rubber (LSR) is processed through injection molding, allowing for extremely precise and complex geometries. In contrast, solid silicone rubber is often processed through compression molding for simpler, high-volume parts. Here is the bottom line: LSR provides superior clarity and purity, making it the gold standard for medical and infant care applications.
- LSR: High precision, rapid cycle times.
- Solid Rubber: Cost-effective for gaskets.
- Gel: Vibration damping and cushioning.
- Resin: Rigid coatings and insulation.
Why consistencies vary across industrial applications
The versatility of silicone allows you to specify everything from a soft, skin-like texture to a hard, plastic-like shell. By manipulating the filler materials and curing parameters, manufacturers can tailor the material’s tensile strength and elongation. This adaptability ensures that silicone can replace both soft rubbers and rigid plastics in your supply chain.
Key Takeaway: Silicone rubber is the high-performance elastomeric version of the silicone polymer, optimized for mechanical durability and shape retention.
| Form | Typical Use Case | Key Advantage |
|---|---|---|
| Liquid (LSR) | Medical Valves | High Purity |
| Solid (HTV) | Automotive Seals | Thermal Stability |
| Gels | Electronics Potting | Shock Absorption |
| Fluids | Industrial Lubricants | Viscosity Stability |
Tailoring the consistency of silicone ensures it meets the specific mechanical demands of your project.
Is silicone a safer alternative to conventional polymers?

Silicone is widely considered a safer alternative to conventional polymers because it is biocompatible, hypoallergenic, and does not leach harmful chemical additives into its environment. Concerns regarding microplastics from silicone are minimal because the material remains stable and does not degrade into the toxic endocrine disruptors associated with traditional plastics. You can trust silicone for food-contact and medical applications where material purity is the primary concern. Its ability to withstand sterilization and high-heat environments without breaking down further cements its reputation as a premium safety material.
Biocompatibility and hypoallergenic properties
Silicone does not react with human tissue or biological fluids, making it the preferred material for long-term implants and skin-contact devices. If you are developing products for sensitive users, such as infants or medical patients, silicone’s hypoallergenic nature reduces the risk of allergic reactions. This safety profile is unmatched by most thermoplastic elastomers or vinyl-based materials.
Why silicone does not leach harmful chemicals
Traditional plastics often contain “free” molecules like plasticizers that can migrate out of the material over time, especially when heated. Silicone is a thermoset material with a stable cured structure that locks its components in place. But it gets better: because it requires no heavy metal stabilizers or harmful catalysts (when platinum-cured), the risk of contamination is virtually zero.
- No Bisphenol-A (BPA).
- Phthalate-free construction.
- Lead and latex-free.
- FDA and LFGB compliant grades available.
Regulatory standards for food-grade materials
Food-grade silicone must pass rigorous testing to ensure it does not impart flavor or odor to food products. These standards ensure that the material can be used repeatedly in ovens, dishwashers, and freezers without losing its structural integrity. When you source certified food-grade silicone, you are investing in a product that prioritizes consumer health and long-term reliability.
Key Takeaway: Silicone’s inherent chemical stability and lack of toxic additives make it the safest polymer choice for health-conscious and regulated industries.
| Factor | Silicone | PVC/Vinyl |
|---|---|---|
| Plasticizers | None Required | High Levels |
| Heat Leaching | Negligible | Significant |
| Allergen Risk | Hypoallergenic | Possible Latex/Dye issues |
| Recyclability | Specialized | Difficult/Toxic |
The absence of harmful leaching makes silicone the superior choice for high-heat and health-sensitive products.
Does silicone produce microplastics from silicone over time?
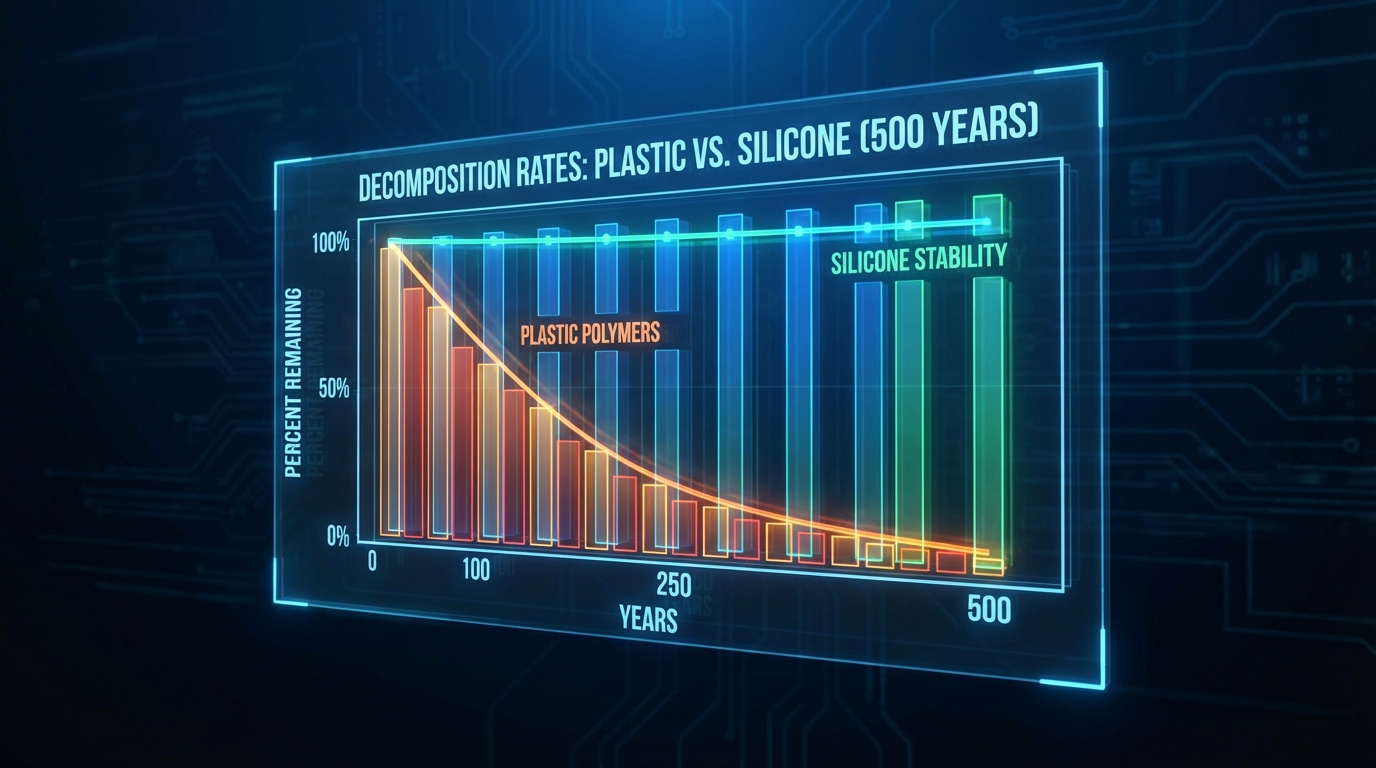
Silicone does not produce microplastics in the traditional sense because it is not a plastic; however, it can break down into siloxane fragments if subjected to extreme mechanical or chemical stress. These fragments of microplastics from silicone are chemically inert and do not behave like the persistent, bioaccumulative toxic particles derived from petroleum polymers. You will find that while plastic fragments attract and carry heavy metals in the ocean, siloxane degradation products tend to be much more benign. The breakdown process is incredibly slow, often taking centuries, which contributes to the material’s reputation for extreme longevity.
Analyzing the breakdown process of siloxane
The degradation of silicone occurs primarily through hydrolysis or oxidation at extremely high temperatures rather than the simple mechanical fragmentation seen in plastics. This means the material maintains its macro-structure much longer than a plastic bag or bottle would. You can observe in the decomposition charts that silicone’s stability curve remains nearly flat for hundreds of years compared to the rapid decay of plastics.
Why silicone is chemically inert in the environment
Because silicone is inorganic, it does not provide a food source for bacteria or fungi, which prevents biological degradation. This inertness means that even if a piece of silicone ends up in a landfill, it will stay there as a solid piece rather than leaching chemicals into the groundwater. You are essentially dealing with a man-made stone rather than a reactive chemical pollutant.
Here is the bottom line:
- Minimal fragmentation into tiny particles.
- No attraction of persistent organic pollutants (POPs).
- Degradation products are primarily silica and CO2.
- Extremely high resistance to environmental weathering.
Distinguishing silicone degradation from plastic fragmentation
Plastic fragmentation results in billions of microscopic beads that enter the food chain and human bloodstream. Silicone, due to its higher surface energy and internal bonding, does not easily “dust” or flake off. This physical integrity is why you can use silicone products for years without them becoming thin, brittle, or powdery.
Key Takeaway: Silicone is an environmentally stable material that avoids the toxic microplastic cycle, making it a responsible choice for long-term product lifecycles.
| Breakdown Factor | Silicone | Polyethylene (Plastic) |
|---|---|---|
| Primary Mechanism | Slow Hydrolysis | Photo-degradation/Fragmentation |
| Particle Toxicity | Inert Siloxanes | Toxic Microplastics |
| Environmental Half-life | 500+ Years | 50-100 Years (to micro-stage) |
| Bioaccumulation | Non-existent | High |
The data confirms that silicone remains structurally intact far longer than traditional plastics, preventing particle pollution.
What makes silicone superior in extreme temperatures?

Silicone’s superiority in extreme temperatures stems from the high bond energy of the silicon-oxygen backbone, which resists thermal agitation that would melt or freeze traditional polymers. Even in the presence of microplastics from silicone degradation at extreme limits, the material retains its mechanical properties where others turn brittle or liquid. You can rely on silicone to function perfectly at temperatures as low as -60°C and as high as 250°C. This massive operating window makes it an essential material for the most demanding engineering environments on the planet.
Thermal stability from freezing to high heat
While many rubbers become hard and crack in the cold, silicone stays flexible, maintaining its sealing power in arctic conditions. Conversely, in the heat of an industrial oven or an engine bay, silicone does not soften or lose its shape. This reliability allows you to design products that can transition from a deep freezer directly into a high-heat sterilization cycle without failure.
Applications in aerospace and automotive industries
In aerospace, silicone is used for airframe seals and gaskets where it must survive the extreme cold of high altitudes and the intense heat of engine proximity. Automotive engineers utilize it for turbocharger hoses and spark plug boots because it can handle the constant thermal cycling of a modern engine. But it gets better: silicone also provides excellent electrical insulation, which remains stable even as temperatures fluctuate wildly.
- Resists thermal expansion and contraction.
- Maintains dielectric strength at high heat.
- Prevents gasket “set” or permanent deformation.
- Outperforms EPDM and Nitrile in heat resistance.
Why silicone retains elasticity in cold environments
The large atomic size of silicon and the wide bond angles in the siloxane chain allow for significant molecular movement even at low energy levels. This means the polymer chains don’t “lock” together as easily as the tight, carbon-based chains in plastics. You can feel this difference in the soft, pliable nature of silicone even when it is pulled from a commercial freezer.
Key Takeaway: Silicone’s unmatched thermal range makes it the only viable choice for applications that span the extreme ends of the temperature spectrum.
| Environment | Silicone Performance | Standard Rubber Performance |
|---|---|---|
| Cryogenic (-60°C) | Remains Flexible | Brittle/Cracks |
| Boiling Water | No Change | Potential Softening |
| High Heat (200°C+) | Stable for 10,000+ hours | Rapid Melting/Failure |
| Fire Exposure | Self-extinguishing/Non-toxic ash | Toxic Fumes/Melting |
Silicone provides a safety margin in temperature-critical applications that other materials simply cannot match.
How do microplastics from silicone impact the environment?

The impact of microplastics from silicone on the environment is significantly lower than that of plastic because siloxane particles do not biodegrade into harmful chemicals or bioaccumulate in marine life. Because silicone is essentially a synthetic cousin of quartz sand, it remains chemically inert and does not release the endocrine-disrupting gases that plastics emit. You are choosing a material that, while persistent, acts as a “passive” waste rather than an “active” pollutant. Its extreme durability means that one silicone product can replace hundreds of single-use plastic items, drastically reducing the total volume of waste generated.
The non-biodegradable nature of synthetic siloxanes
While non-biodegradability is often seen as a negative, in the case of silicone, it prevents the release of micro-particles into the ecosystem. The material does not rot, mold, or break down into sludge, ensuring that it doesn’t contaminate soil or water supplies. You can view silicone as a long-term storage medium for the resources used to create it, rather than a fleeting consumer good destined for the ocean.
Comparing silicone waste to traditional plastic pollution
Plastic waste often breaks down into “nurdles” and micro-particles that are mistaken for food by wildlife, causing systemic ecological collapse. Silicone waste tends to stay in larger, identifiable pieces that do not easily enter the food chain. Here is the bottom line: while we must still manage silicone waste responsibly, it does not pose the same urgent “microplastic crisis” as polyethylene or polypropylene.
- Reduces reliance on single-use plastics.
- Can be downcycled into industrial oils or lubricants.
- Does not release methane during decomposition.
- Inert in landfills; does not leach into groundwater.
The role of durability in waste reduction
The most effective way you can protect the environment is by choosing materials that don’t need to be replaced. A single silicone food storage bag can be washed and reused thousands of times, preventing the disposal of thousands of plastic sandwich bags. This “buy once, use forever” mentality is the core of silicone’s environmental advantage in the modern economy.
Key Takeaway: Silicone’s permanence and inertness make it a sustainable alternative to the high-turnover, high-pollution lifecycle of traditional plastics.
| Impact Category | Silicone | Single-Use Plastic |
|---|---|---|
| Lifecycle Duration | Years/Decades | Minutes/Hours |
| Ocean Toxicity | Low (Inert) | High (Toxic Particles) |
| Wildlife Ingestion | Low Risk | Extremely High Risk |
| Resource Efficiency | High (due to longevity) | Low (linear economy) |
Choosing durable silicone is a strategic move toward a circular economy and reduced environmental toxicity.
What are the main limitations of using silicone?

The main limitations of silicone include its higher production cost, lower tensile strength compared to some high-performance plastics, and the need for specialized recycling facilities to manage microplastics from silicone waste. You will find that while silicone is incredibly flexible, it can be susceptible to tearing once a small notch or cut is introduced to the surface. This “nick sensitivity” means you must design silicone parts with smooth radii and avoid sharp edges that could act as stress concentrators. Despite these challenges, the functional benefits of silicone often outweigh the costs for specialized applications.
Evaluating the higher cost of production
Silicone production is an energy-intensive process that involves complex chemical synthesis and expensive catalysts like platinum. This results in a raw material price that can be several times higher than that of common plastics like PVC or PE. When you are budgeting for a project, you must account for this premium by considering the total cost of ownership and the reduced need for replacements.
Understanding low tensile strength constraints
Silicone is not a structural material; it cannot support heavy loads or resist high-pressure shearing as well as nylon or polycarbonate. If your application requires rigid support or extreme pull-strength, you may need to overmold silicone onto a metal or plastic substrate. But it gets better: by combining materials, you can enjoy the soft-touch benefits of silicone without sacrificing the structural integrity of the final assembly.
- Susceptible to swelling when exposed to certain oils.
- Higher gas permeability than some plastics.
- Difficult to bond or paint without primers.
- Limited color vibrancy compared to some thermoplastics.
Why specialized recycling is required
Because silicone is a thermoset material (it doesn’t melt once cured), it cannot be simply chopped up and melted down like a plastic bottle. It requires a chemical process to break the siloxane bonds or a mechanical process to turn it into “re-grind” filler for new products. You must partner with specialized recyclers to ensure that end-of-life silicone is handled in an eco-friendly manner.
Key Takeaway: Acknowledging silicone’s mechanical and economic limits allows you to design more effective products that leverage its unique strengths.
| Limitation | Impact on Design | Mitigation Strategy |
|---|---|---|
| Tear Strength | High Nick Sensitivity | Use radiused edges/thicker walls |
| Cost | Higher Initial Investment | Emphasize durability/longevity |
| Bonding | Poor Adhesion | Use plasma treatment or primers |
| Recycling | Not Kerbside Compatible | Use dedicated take-back programs |
Careful engineering can overcome most of silicone’s inherent physical limitations in high-performance environments.
How is medical grade silicone used in healthcare?

Medical grade silicone is used in healthcare for critical applications like surgical implants, tubing, and prosthetic devices because of its unparalleled biocompatibility and resistance to bacterial growth. When sourcing medical grade silicone, you are choosing a material that has undergone stringent testing to ensure it does not cause inflammation or rejection within the body. It is also the preferred choice for baby products such as pacifiers and bottle nipples due to its non-toxic, tasteless, and odorless properties. The material’s ability to withstand repeated steam autoclaving makes it indispensable in sterile hospital environments.
Standards for implants and surgical instruments
Silicone used for permanent implants must meet USP Class VI standards, which involve rigorous biological reactivity tests. This ensures that the material can sit inside the human body for years without degrading or causing a systemic immune response. You will see silicone used in everything from heart valve components to aesthetic implants because no other material provides the same combination of softness and biological inertness.
Benefits for baby products and maternal care
For new parents, the safety of silicone is a primary selling point over traditional latex or plastic. Silicone does not contain nitrosamines or other potential carcinogens found in some rubbers, and it is far more durable than latex, which can become sticky and degrade. Here is the bottom line: silicone provides the safest possible interface for infant feeding and soothing.
- Latex-free and hypoallergenic.
- Bite-resistant and highly durable.
- Transparent grades allow for visual inspection.
- Dishwasher and sterilizer safe.
Why biocompatibility is essential for medical safety
Biocompatibility means the material does not “talk” to the body’s cells, preventing the triggering of the clotting cascade or inflammatory pathways. In catheters and long-term tubing, this prevents the buildup of biofilms and reduces the risk of hospital-acquired infections. You are not just buying a tube; you are buying a safety barrier that protects the patient’s health.
Key Takeaway: The extreme purity and biological safety of medical-grade silicone make it the foundation of modern surgical and infant care technology.
| Application | Key Requirement | Silicone Benefit |
|---|---|---|
| Implants | Long-term Inertness | No tissue reaction |
| Surgical Tubing | Kink Resistance | Maintains flow/biocompatible |
| Pacifiers | Non-toxicity | No leaching/Latex-free |
| Gaskets/Seals | Sterilizability | Withstands 134°C Autoclave |
The medical field relies on silicone’s unique ability to coexist with human biology without adverse effects.
Where do we encounter silicone in our daily lives?

You encounter silicone daily in everything from the non-stick bakeware in your kitchen to the protective sleeves that shield your high-end electronics from damage. In the tech industry, smart wearable accessories like watch straps utilize silicone because it is sweat-resistant, comfortable on the skin, and does not shed microplastics from silicone during vigorous activity. Its presence is often invisible, acting as the sealant around your windows or the lubricant in your personal care products, yet its impact on modern convenience is profound. From the car you drive to the phone in your pocket, silicone is the silent partner providing durability and protection.
Food-grade kitchenware and cookware essentials
In the modern kitchen, silicone has replaced traditional metal and wood for many tasks due to its heat resistance and non-stick nature. You can use silicone spatulas to scrape hot pans without melting them, and silicone baking mats eliminate the need for disposable parchment paper. This transition not only improves cooking efficiency but also reduces the chemical exposure associated with older non-stick coatings.
Smart wearable accessories and acoustic components
The wearable market relies on silicone for its unique “soft touch” and durability. It provides a secure, comfortable fit for fitness trackers that must be worn 24/7, resisting the oils and salts of human skin. But it gets better: in the world of audio, silicone ear-tips provide a perfect seal for noise isolation, ensuring that high-fidelity sound is delivered directly to your ears without external interference.
- Shock-absorbing phone cases.
- Waterproof seals for smartwatches.
- Ergonomic grips for kitchen tools.
- Soft-touch buttons for remote controls.
Why appearance and texture matter
In consumer electronics, the tactile feel of a product is as important as its function. Silicone allows designers to create surfaces that feel premium, organic, and slip-resistant. You can choose from a variety of finishes, from matte to high-gloss, and customize the color to match any brand identity, ensuring that functional protection never compromises on style.
Key Takeaway: Silicone is the versatile “super-material” of the consumer world, blending industrial-grade protection with everyday comfort and style.
| Daily Item | Primary Function | Why Silicone? |
|---|---|---|
| Phone Case | Drop Protection | Impact absorption/Soft grip |
| Watch Strap | Skin Contact | Sweat-proof/Hypoallergenic |
| Baking Mold | High Heat Cooking | Non-stick/Flexible |
| Window Sealant | Weatherproofing | UV stable/Waterproof |
Silicone’s pervasive nature in modern life is a testament to its unmatched balance of safety, durability, and versatility.
Conclusion
The journey of understanding high-performance materials reveals that while silicone may visually resemble plastic, its chemical soul is rooted in the stability of silica sand. We have explored how its unique siloxane backbone prevents the environmental crisis of microplastics while providing a thermal and chemical range that traditional polymers cannot match. From the sterile environments of surgical theaters to the high-heat demands of professional kitchens, silicone stands as a beacon of safety and durability. Whether you are seeking to optimize your industrial supply chain or looking for safer consumer products, the evidence points toward silicone as the most responsible and effective choice.
If you are ready to elevate your product standards and move away from the limitations of traditional plastics, contact us today. We have spent over 15 years mastering the intricacies of LSR and custom silicone manufacturing to solve the most complex engineering challenges. Our team can help you transition to biocompatible, high-durability solutions that protect both your brand reputation and the environment. Let’s build a future where performance and sustainability coexist through the power of precision-engineered silicone.
FAQ
Can I recycle silicone at home?
No, silicone cannot be recycled through your standard curbside bin because it requires specialized chemical processes to break down its thermoset structure. You should look for dedicated take-back programs or industrial recyclers who can grind the material for use in playground surfaces or convert it back into silicone oils.
What’s the best way to clean white silicone shoes?
The best method involves using a mixture of mild soap and warm water with a soft-bristled brush to gently lift surface contaminants. Avoid using harsh bleach or abrasive chemicals, as high-quality silicone is naturally stain-resistant and usually only requires a gentle physical scrub to restore its original appearance.
Is silicone bad for babies?
No, silicone is generally considered the safest material for baby products because it is non-toxic, hypoallergenic, and free from BPA, phthalates, and heavy metals. Medical-grade silicone’s resistance to heat also means it can be thoroughly sterilized without any risk of chemical leaching or structural degradation.
Does silicone turn yellow over time?
It depends on the quality of the material and its exposure to specific environmental factors like high heat or intense UV radiation. While high-grade silicone is much more resistant to oxidation than plastic, low-quality additives in cheaper versions can cause slight discoloration over many years of use.
Is silicone more expensive than plastic?
Yes, the raw material and manufacturing costs for silicone are significantly higher due to the complex chemical synthesis and energy required for processing. However, the investment is offset by the material’s extreme longevity, as one silicone part can often outlast five to ten plastic equivalents in demanding environments.
